| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Protease |
|---|
| Ligand | BDBM50061306 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_469686 (CHEMBL946814) |
|---|
| Ki | 0.931±n/a nM |
|---|
| Citation |  Mimoto, T; Nojima, S; Terashima, K; Takaku, H; Shintani, M; Hayashi, H Structure-activity relationships of novel HIV-1 protease inhibitors containing the 3-amino-2-chlorobenzoyl-allophenylnorstatine structure. Bioorg Med Chem16:1299-308 (2008) [PubMed] Article Mimoto, T; Nojima, S; Terashima, K; Takaku, H; Shintani, M; Hayashi, H Structure-activity relationships of novel HIV-1 protease inhibitors containing the 3-amino-2-chlorobenzoyl-allophenylnorstatine structure. Bioorg Med Chem16:1299-308 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Protease |
|---|
| Name: | Protease |
|---|
| Synonyms: | n/a |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 10904.79 |
|---|
| Organism: | Human immunodeficiency virus 1 (HIV-1) |
|---|
| Description: | Q9YQ12 |
|---|
| Residue: | 99 |
|---|
| Sequence: | PQITLWQRPFVTIKIEGQLKEALLDTGADDTVLEEMNLPGRWKPKMIGGIGGFIKVRQYD
QIVIEICGKKAIGTVLVGPTPVNIIGRNLLTQIGCTLNF
|
|
|
|---|
| BDBM50061306 |
|---|
| n/a |
|---|
| Name | BDBM50061306 |
|---|
| Synonyms: | (3S,4aS,8aS)-2-[(2R,3R)-2-Hydroxy-3-(3-hydroxy-2-methyl-benzoylamino)-4-phenylsulfanyl-butyl]-decahydro-isoquinoline-3-carboxylic acid tert-butylamide; compound with methanesulfonic acid | AG-1343 | CHEMBL1205 | NELFINAVIR MESYLATE | Nelfinavir | Viracept | cmdc.202100576, 24h |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H45N3O4S |
|---|
| Mol. Mass. | 567.782 |
|---|
| SMILES | Cc1c(O)cccc1C(=O)N[C@@H](CSc1ccccc1)[C@H](O)CN1C[C@H]2CCCC[C@H]2C[C@H]1C(=O)NC(C)(C)C |r| |
|---|
| Structure | 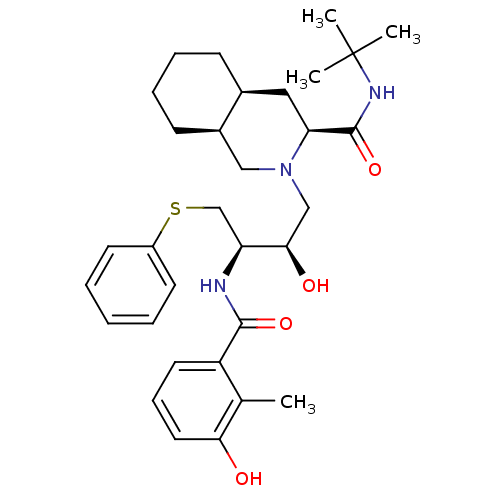
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mimoto, T; Nojima, S; Terashima, K; Takaku, H; Shintani, M; Hayashi, H Structure-activity relationships of novel HIV-1 protease inhibitors containing the 3-amino-2-chlorobenzoyl-allophenylnorstatine structure. Bioorg Med Chem16:1299-308 (2008) [PubMed] Article
Mimoto, T; Nojima, S; Terashima, K; Takaku, H; Shintani, M; Hayashi, H Structure-activity relationships of novel HIV-1 protease inhibitors containing the 3-amino-2-chlorobenzoyl-allophenylnorstatine structure. Bioorg Med Chem16:1299-308 (2008) [PubMed] Article