| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 1,3-beta-D-glucan synthase catalytic subunit |
|---|
| Ligand | BDBM50478215 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_683511 (CHEMBL1286426) |
|---|
| IC50 | 0.500000±n/a nM |
|---|
| Citation |  Garcia-Effron, G; Kontoyiannis, DP; Lewis, RE; Perlin, DS Caspofungin-resistant Candida tropicalis strains causing breakthrough fungemia in patients at high risk for hematologic malignancies. Antimicrob Agents Chemother52:4181-3 (2008) [PubMed] Article Garcia-Effron, G; Kontoyiannis, DP; Lewis, RE; Perlin, DS Caspofungin-resistant Candida tropicalis strains causing breakthrough fungemia in patients at high risk for hematologic malignancies. Antimicrob Agents Chemother52:4181-3 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 1,3-beta-D-glucan synthase catalytic subunit |
|---|
| Name: | 1,3-beta-D-glucan synthase catalytic subunit |
|---|
| Synonyms: | 1,3-beta-D-glucan synthase catalytic subunit | Beta-1,3-glucan synthase |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 80469.77 |
|---|
| Organism: | Candida albicans |
|---|
| Description: | ChEMBL_663643 |
|---|
| Residue: | 690 |
|---|
| Sequence: | GTIYWMYTAYNSPTLYTKHYVQTINQQPLASSRWAACAIGGVLASFIQILATLFEWIFVP
REWAGAQHLSRRMLFLVLIFLLNLVPPVYTFQITKLVIYSKSAYAVSIVGFFIAVATLVF
FAVMPLGGLFTSYMNKRSRRYIASQTFTANYIKLKGLDMWMSYLLWFLVFLAKLVESYFF
LTLSLRDPIRNLSTMTMRCVGEVWYKDIVCRNQAKIVLGLMYLVDLLLFFLDTYMWYIIC
NCIFSIGRSFYLGISILTPWRNIFTRLPKRIYSKILATTEMEIKYKPKVLISQIWNAIVI
SMYREHLLAIDHVQKLLYHQVPSEIEGKRTLRAPTFFVSQDDNNFETEFFPRNSEAERRI
SFFAQSLATPMPEPLPVDNMPTFTVFTPHYSEKILLSLREIIREDDQFSRVTLLEYLKQL
HPVEWDCFVKDTKILAEETAAYENGDDSEKLSEDGLKSKIDDLPFYCIGFKSAAPEYTLR
TRIWASLRSQTLYRTVSGFMNYARAIKLLYRVENPELVQYFGGDPEGLELALERMARRKF
RFLVSMQRLSKFKDDEMENAEFLLRAYPDLQIAYLDEEPALNEDEEPRVYSALIDGHCEM
LENGRRRPKFRVQLSGNPILGDGKSDNQNHAVIFHRGEYIQLIDANQDNYLEECLKIRSV
LAEFEEMNVEHVNPYAPNLKSEDNNTKKDP
|
|
|
|---|
| BDBM50478215 |
|---|
| n/a |
|---|
| Name | BDBM50478215 |
|---|
| Synonyms: | CHEBI:474180 | Cancidas | Caspofungin | Caspofungin Acetate | L-743,872 | L-743872 | MK-991 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C52H88N10O15 |
|---|
| Mol. Mass. | 1093.3131 |
|---|
| SMILES | [H][C@@]12C[C@@H](O)CN1C(=O)[C@@]([H])(NC(=O)[C@H](C[C@@H](O)[C@@H](NCCN)NC(=O)[C@]1([H])[C@@H](O)CCN1C(=O)[C@@]([H])(NC(=O)[C@@]([H])(NC2=O)[C@H](O)[C@@H](O)c1ccc(O)cc1)[C@H](O)CCN)NC(=O)CCCCCCCCC(C)CC(C)CC)[C@@H](C)O |r| |
|---|
| Structure | 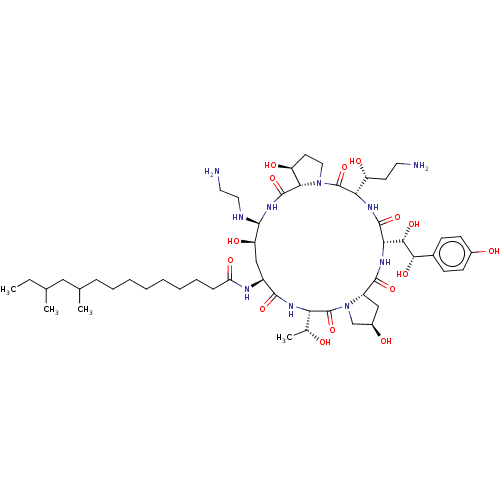
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Garcia-Effron, G; Kontoyiannis, DP; Lewis, RE; Perlin, DS Caspofungin-resistant Candida tropicalis strains causing breakthrough fungemia in patients at high risk for hematologic malignancies. Antimicrob Agents Chemother52:4181-3 (2008) [PubMed] Article
Garcia-Effron, G; Kontoyiannis, DP; Lewis, RE; Perlin, DS Caspofungin-resistant Candida tropicalis strains causing breakthrough fungemia in patients at high risk for hematologic malignancies. Antimicrob Agents Chemother52:4181-3 (2008) [PubMed] Article