| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prolyl endopeptidase |
|---|
| Ligand | BDBM50393842 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1836618 (CHEMBL4336751) |
|---|
| Ki | 160±n/a nM |
|---|
| Citation |  Plescia, J; De Cesco, S; Patrascu, MB; Kurian, J; Di Trani, J; Dufresne, C; Wahba, AS; Janmamode, N; Mittermaier, AK; Moitessier, N Integrated Synthetic, Biophysical, and Computational Investigations of Covalent Inhibitors of Prolyl Oligopeptidase and Fibroblast Activation Protein ?. J Med Chem62:7874-7884 (2019) [PubMed] Article Plescia, J; De Cesco, S; Patrascu, MB; Kurian, J; Di Trani, J; Dufresne, C; Wahba, AS; Janmamode, N; Mittermaier, AK; Moitessier, N Integrated Synthetic, Biophysical, and Computational Investigations of Covalent Inhibitors of Prolyl Oligopeptidase and Fibroblast Activation Protein ?. J Med Chem62:7874-7884 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prolyl endopeptidase |
|---|
| Name: | Prolyl endopeptidase |
|---|
| Synonyms: | PE | PEP | POP | PPCE_HUMAN | PREP | Post-proline cleaving enzyme | Prolyl oligopeptidase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 80688.50 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P48147 |
|---|
| Residue: | 710 |
|---|
| Sequence: | MLSLQYPDVYRDETAVQDYHGHKICDPYAWLEDPDSEQTKAFVEAQNKITVPFLEQCPIR
GLYKERMTELYDYPKYSCHFKKGKRYFYFYNTGLQNQRVLYVQDSLEGEARVFLDPNILS
DDGTVALRGYAFSEDGEYFAYGLSASGSDWVTIKFMKVDGAKELPDVLERVKFSCMAWTH
DGKGMFYNSYPQQDGKSDGTETSTNLHQKLYYHVLGTDQSEDILCAEFPDEPKWMGGAEL
SDDGRYVLLSIREGCDPVNRLWYCDLQQESSGIAGILKWVKLIDNFEGEYDYVTNEGTVF
TFKTNRQSPNYRVINIDFRDPEESKWKVLVPEHEKDVLEWIACVRSNFLVLCYLHDVKNI
LQLHDLTTGALLKTFPLDVGSIVGYSGQKKDTEIFYQFTSFLSPGIIYHCDLTKEELEPR
VFREVTVKGIDASDYQTVQIFYPSKDGTKIPMFIVHKKGIKLDGSHPAFLYGYGGFNISI
TPNYSVSRLIFVRHMGGILAVANIRGGGEYGETWHKGGILANKQNCFDDFQCAAEYLIKE
GYTSPKRLTINGGSNGGLLVAACANQRPDLFGCVIAQVGVMDMLKFHKYTIGHAWTTDYG
CSDSKQHFEWLVKYSPLHNVKLPEADDIQYPSMLLLTADHDDRVVPLHSLKFIATLQYIV
GRSRKQSNPLLIHVDTKAGHGAGKPTAKVIEEVSDMFAFIARCLNVDWIP
|
|
|
|---|
| BDBM50393842 |
|---|
| n/a |
|---|
| Name | BDBM50393842 |
|---|
| Synonyms: | CHEMBL2159749 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H20N2O2 |
|---|
| Mol. Mass. | 320.385 |
|---|
| SMILES | O=C(N1CCCC1)c1cccc2CN(Cc3ccccc3)C(=O)c12 |
|---|
| Structure | 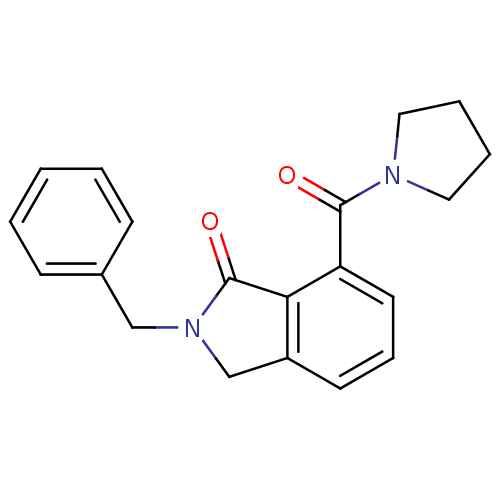
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Plescia, J; De Cesco, S; Patrascu, MB; Kurian, J; Di Trani, J; Dufresne, C; Wahba, AS; Janmamode, N; Mittermaier, AK; Moitessier, N Integrated Synthetic, Biophysical, and Computational Investigations of Covalent Inhibitors of Prolyl Oligopeptidase and Fibroblast Activation Protein ?. J Med Chem62:7874-7884 (2019) [PubMed] Article
Plescia, J; De Cesco, S; Patrascu, MB; Kurian, J; Di Trani, J; Dufresne, C; Wahba, AS; Janmamode, N; Mittermaier, AK; Moitessier, N Integrated Synthetic, Biophysical, and Computational Investigations of Covalent Inhibitors of Prolyl Oligopeptidase and Fibroblast Activation Protein ?. J Med Chem62:7874-7884 (2019) [PubMed] Article