| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 4B1 |
|---|
| Ligand | BDBM330295 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2078100 (CHEMBL4733891) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Xiang, HY; Wang, X; Chen, YH; Zhang, X; Tan, C; Wang, Y; Su, Y; Gao, ZW; Chen, XY; Xiong, B; Gao, ZB; Chen, Y; Ding, J; Meng, LH; Yang, CH Identification of methyl (5-(6-((4-(methylsulfonyl)piperazin-1-yl)methyl)-4-morpholinopyrrolo[2,1-f][1,2,4]triazin-2-yl)-4-(trifluoromethyl)pyridin-2-yl)carbamate (CYH33) as an orally bioavailable, highly potent, PI3K alpha inhibitor for the treatment of advanced solid tumors. Eur J Med Chem209:0 (2021) [PubMed] Article Xiang, HY; Wang, X; Chen, YH; Zhang, X; Tan, C; Wang, Y; Su, Y; Gao, ZW; Chen, XY; Xiong, B; Gao, ZB; Chen, Y; Ding, J; Meng, LH; Yang, CH Identification of methyl (5-(6-((4-(methylsulfonyl)piperazin-1-yl)methyl)-4-morpholinopyrrolo[2,1-f][1,2,4]triazin-2-yl)-4-(trifluoromethyl)pyridin-2-yl)carbamate (CYH33) as an orally bioavailable, highly potent, PI3K alpha inhibitor for the treatment of advanced solid tumors. Eur J Med Chem209:0 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 4B1 |
|---|
| Name: | Cytochrome P450 4B1 |
|---|
| Synonyms: | 1.14.14.1 | CP4B1_HUMAN | CYP4B1 | CYPIVB1 | Cytochrome P450 4B1 | Cytochrome P450-HP |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 59005.59 |
|---|
| Organism: | Homo sapiens |
|---|
| Description: | ChEMBL_119703 |
|---|
| Residue: | 511 |
|---|
| Sequence: | MVPSFLSLSFSSLGLWASGLILVLGFLKLIHLLLRRQTLAKAMDKFPGPPTHWLFGHALE
IQETGSLDKVVSWAHQFPYAHPLWFGQFIGFLNIYEPDYAKAVYSRGDPKAPDVYDFFLQ
WIGRGLLVLEGPKWLQHRKLLTPGFHYDVLKPYVAVFTESTRIMLDKWEEKAREGKSFDI
FCDVGHMALNTLMKCTFGRGDTGLGHRDSSYYLAVSDLTLLMQQRLVSFQYHNDFIYWLT
PHGRRFLRACQVAHDHTDQVIRERKAALQDEKVRKKIQNRRHLDFLDILLGARDEDDIKL
SDADLRAEVDTFMFEGHDTTTSGISWFLYCMALYPEHQHRCREEVREILGDQDFFQWDDL
GKMTYLTMCIKESFRLYPPVPQVYRQLSKPVTFVDGRSLPAGSLISMHIYALHRNSAVWP
DPEVFDSLRFSTENASKRHPFAFMPFSAGPRNCIGQQFAMSEMKVVTAMCLLRFEFSLDP
SRLPIKMPQLVLRSKNGFHLHLKPLGPGSGK
|
|
|
|---|
| BDBM330295 |
|---|
| n/a |
|---|
| Name | BDBM330295 |
|---|
| Synonyms: | Methyl (5-(6-((4-(methylsulfonyl)piperazin-1-yl)methyl)-4-morpholinopyrrolo[2,1-f][1,2,4]triazin-2-yl)-4-(trifluoromethyl)pyridin-2-yl)carbamate | US9724352, I-33 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H29F3N8O5S |
|---|
| Mol. Mass. | 598.598 |
|---|
| SMILES | COC(=O)Nc1cc(c(cn1)-c1nc(N2CCOCC2)c2cc(CN3CCN(CC3)S(C)(=O)=O)cn2n1)C(F)(F)F |
|---|
| Structure | 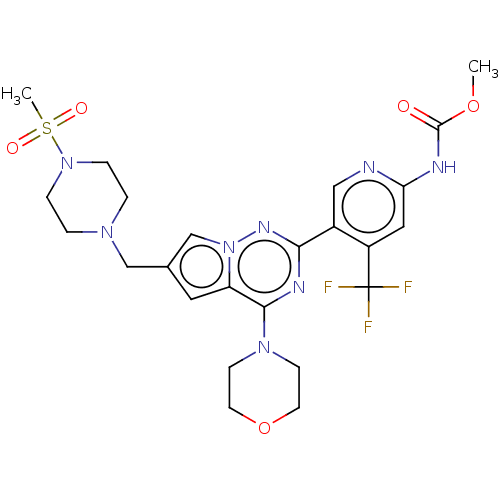
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Xiang, HY; Wang, X; Chen, YH; Zhang, X; Tan, C; Wang, Y; Su, Y; Gao, ZW; Chen, XY; Xiong, B; Gao, ZB; Chen, Y; Ding, J; Meng, LH; Yang, CH Identification of methyl (5-(6-((4-(methylsulfonyl)piperazin-1-yl)methyl)-4-morpholinopyrrolo[2,1-f][1,2,4]triazin-2-yl)-4-(trifluoromethyl)pyridin-2-yl)carbamate (CYH33) as an orally bioavailable, highly potent, PI3K alpha inhibitor for the treatment of advanced solid tumors. Eur J Med Chem209:0 (2021) [PubMed] Article
Xiang, HY; Wang, X; Chen, YH; Zhang, X; Tan, C; Wang, Y; Su, Y; Gao, ZW; Chen, XY; Xiong, B; Gao, ZB; Chen, Y; Ding, J; Meng, LH; Yang, CH Identification of methyl (5-(6-((4-(methylsulfonyl)piperazin-1-yl)methyl)-4-morpholinopyrrolo[2,1-f][1,2,4]triazin-2-yl)-4-(trifluoromethyl)pyridin-2-yl)carbamate (CYH33) as an orally bioavailable, highly potent, PI3K alpha inhibitor for the treatment of advanced solid tumors. Eur J Med Chem209:0 (2021) [PubMed] Article