| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50583947 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2158480 (CHEMBL5043230) |
|---|
| IC50 | 2000±n/a nM |
|---|
| Citation |  Hopkins, BT; Bame, E; Bajrami, B; Black, C; Bohnert, T; Boiselle, C; Burdette, D; Burns, JC; Delva, L; Donaldson, D; Grater, R; Gu, C; Hoemberger, M; Johnson, J; Kapadnis, S; King, K; Lulla, M; Ma, B; Marx, I; Magee, T; Meissner, R; Metrick, CM; Mingueneau, M; Murugan, P; Otipoby, KL; Polack, E; Poreci, U; Prince, R; Roach, AM; Rowbottom, C; Santoro, JC; Schroeder, P; Tang, H; Tien, E; Zhang, F; Lyssikatos, J Discovery and Preclinical Characterization of BIIB091, a Reversible, Selective BTK Inhibitor for the Treatment of Multiple Sclerosis. J Med Chem65:1206-1224 (2022) [PubMed] Article Hopkins, BT; Bame, E; Bajrami, B; Black, C; Bohnert, T; Boiselle, C; Burdette, D; Burns, JC; Delva, L; Donaldson, D; Grater, R; Gu, C; Hoemberger, M; Johnson, J; Kapadnis, S; King, K; Lulla, M; Ma, B; Marx, I; Magee, T; Meissner, R; Metrick, CM; Mingueneau, M; Murugan, P; Otipoby, KL; Polack, E; Poreci, U; Prince, R; Roach, AM; Rowbottom, C; Santoro, JC; Schroeder, P; Tang, H; Tien, E; Zhang, F; Lyssikatos, J Discovery and Preclinical Characterization of BIIB091, a Reversible, Selective BTK Inhibitor for the Treatment of Multiple Sclerosis. J Med Chem65:1206-1224 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50583947 |
|---|
| n/a |
|---|
| Name | BDBM50583947 |
|---|
| Synonyms: | CHEMBL5085931 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H31N9O |
|---|
| Mol. Mass. | 485.584 |
|---|
| SMILES | Cn1cc(Nc2nccc(n2)-c2ccc3[C@@H](CCCCc3c2)NC(=O)c2cn(nn2)C(C)(C)C)cn1 |r| |
|---|
| Structure | 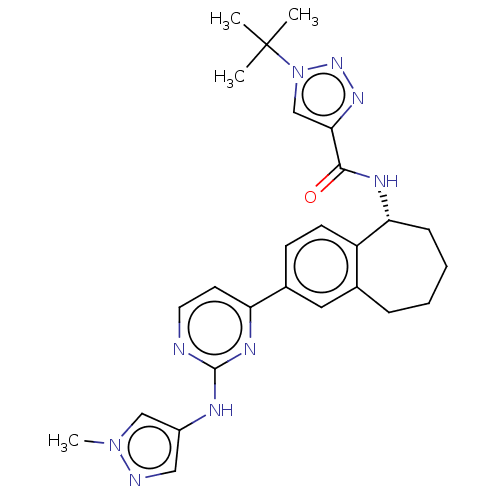
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hopkins, BT; Bame, E; Bajrami, B; Black, C; Bohnert, T; Boiselle, C; Burdette, D; Burns, JC; Delva, L; Donaldson, D; Grater, R; Gu, C; Hoemberger, M; Johnson, J; Kapadnis, S; King, K; Lulla, M; Ma, B; Marx, I; Magee, T; Meissner, R; Metrick, CM; Mingueneau, M; Murugan, P; Otipoby, KL; Polack, E; Poreci, U; Prince, R; Roach, AM; Rowbottom, C; Santoro, JC; Schroeder, P; Tang, H; Tien, E; Zhang, F; Lyssikatos, J Discovery and Preclinical Characterization of BIIB091, a Reversible, Selective BTK Inhibitor for the Treatment of Multiple Sclerosis. J Med Chem65:1206-1224 (2022) [PubMed] Article
Hopkins, BT; Bame, E; Bajrami, B; Black, C; Bohnert, T; Boiselle, C; Burdette, D; Burns, JC; Delva, L; Donaldson, D; Grater, R; Gu, C; Hoemberger, M; Johnson, J; Kapadnis, S; King, K; Lulla, M; Ma, B; Marx, I; Magee, T; Meissner, R; Metrick, CM; Mingueneau, M; Murugan, P; Otipoby, KL; Polack, E; Poreci, U; Prince, R; Roach, AM; Rowbottom, C; Santoro, JC; Schroeder, P; Tang, H; Tien, E; Zhang, F; Lyssikatos, J Discovery and Preclinical Characterization of BIIB091, a Reversible, Selective BTK Inhibitor for the Treatment of Multiple Sclerosis. J Med Chem65:1206-1224 (2022) [PubMed] Article