| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM31772 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2188376 (CHEMBL5100458) |
|---|
| IC50 | 1.000000±n/a nM |
|---|
| Citation |  Fallot, LB; Suresh, RR; Fisher, CL; Salmaso, V; O'Connor, RD; Kaufman, N; Gao, ZG; Auchampach, JA; Jacobson, KA Structure-Activity Studies of 1 J Med Chem65:15238-15262 (2022) [PubMed] Article Fallot, LB; Suresh, RR; Fisher, CL; Salmaso, V; O'Connor, RD; Kaufman, N; Gao, ZG; Auchampach, JA; Jacobson, KA Structure-Activity Studies of 1 J Med Chem65:15238-15262 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM31772 |
|---|
| n/a |
|---|
| Name | BDBM31772 |
|---|
| Synonyms: | 1-[2-(2,4-dichlorobenzyl)oxy-2-(2,4-dichlorophenyl)ethyl]imidazole;nitric acid | Brentan | CHEMBL91 | Dactarin | Miconazole | US9138393, Miconazole Nitrate | US9144538, Miconazole Nitrate | cid_4189 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H14Cl4N2O |
|---|
| Mol. Mass. | 416.129 |
|---|
| SMILES | Clc1ccc(COC(Cn2ccnc2)c2ccc(Cl)cc2Cl)c(Cl)c1 |
|---|
| Structure | 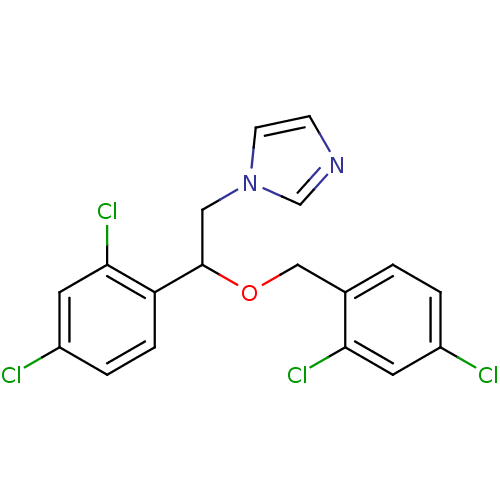
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fallot, LB; Suresh, RR; Fisher, CL; Salmaso, V; O'Connor, RD; Kaufman, N; Gao, ZG; Auchampach, JA; Jacobson, KA Structure-Activity Studies of 1 J Med Chem65:15238-15262 (2022) [PubMed] Article
Fallot, LB; Suresh, RR; Fisher, CL; Salmaso, V; O'Connor, RD; Kaufman, N; Gao, ZG; Auchampach, JA; Jacobson, KA Structure-Activity Studies of 1 J Med Chem65:15238-15262 (2022) [PubMed] Article