| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histamine H1 receptor |
|---|
| Ligand | BDBM50057128 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_303340 (CHEMBL840156) |
|---|
| Ki | >1000±n/a nM |
|---|
| Citation |  Andrés, JI; Alcázar, J; Alonso, JM; Alvarez, RM; Bakker, MH; Biesmans, I; Cid, JM; De Lucas, AI; Fernández, J; Font, LM; Hens, KA; Iturrino, L; Lenaerts, I; Martínez, S; Megens, AA; Pastor, J; Vermote, PC; Steckler, T Discovery of a new series of centrally active tricyclic isoxazoles combining serotonin (5-HT) reuptake inhibition with alpha2-adrenoceptor blocking activity. J Med Chem48:2054-71 (2005) [PubMed] Article Andrés, JI; Alcázar, J; Alonso, JM; Alvarez, RM; Bakker, MH; Biesmans, I; Cid, JM; De Lucas, AI; Fernández, J; Font, LM; Hens, KA; Iturrino, L; Lenaerts, I; Martínez, S; Megens, AA; Pastor, J; Vermote, PC; Steckler, T Discovery of a new series of centrally active tricyclic isoxazoles combining serotonin (5-HT) reuptake inhibition with alpha2-adrenoceptor blocking activity. J Med Chem48:2054-71 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histamine H1 receptor |
|---|
| Name: | Histamine H1 receptor |
|---|
| Synonyms: | H1R | HH1R | HISTAMINE H1 | HRH1 | HRH1_HUMAN |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 55808.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Cell pellets from SK-N-MC cells transfected with human H1 receptor were used in binding assay. |
|---|
| Residue: | 487 |
|---|
| Sequence: | MSLPNSSCLLEDKMCEGNKTTMASPQLMPLVVVLSTICLVTVGLNLLVLYAVRSERKLHT
VGNLYIVSLSVADLIVGAVVMPMNILYLLMSKWSLGRPLCLFWLSMDYVASTASIFSVFI
LCIDRYRSVQQPLRYLKYRTKTRASATILGAWFLSFLWVIPILGWNHFMQQTSVRREDKC
ETDFYDVTWFKVMTAIINFYLPTLLMLWFYAKIYKAVRQHCQHRELINRSLPSFSEIKLR
PENPKGDAKKPGKESPWEVLKRKPKDAGGGSVLKSPSQTPKEMKSPVVFSQEDDREVDKL
YCFPLDIVHMQAAAEGSSRDYVAVNRSHGQLKTDEQGLNTHGASEISEDQMLGDSQSFSR
TDSDTTTETAPGKGKLRSGSNTGLDYIKFTWKRLRSHSRQYVSGLHMNRERKAAKQLGFI
MAAFILCWIPYFIFFMVIAFCKNCCNEHLHMFTIWLGYINSTLNPLIYPLCNENFKKTFK
RILHIRS
|
|
|
|---|
| BDBM50057128 |
|---|
| n/a |
|---|
| Name | BDBM50057128 |
|---|
| Synonyms: | (2-Benzofuran-6-yl-ethyl)-((R)-5-methoxy-1,2,3,4-tetrahydro-naphthalen-1-ylmethyl)-methyl-amine | A-80426 | CHEMBL268258 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H27NO2 |
|---|
| Mol. Mass. | 349.466 |
|---|
| SMILES | COc1cccc2[C@H](CN(C)CCc3ccc4ccoc4c3)CCCc12 |
|---|
| Structure | 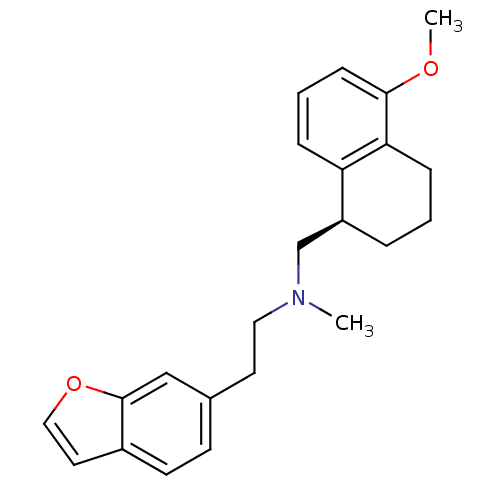
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Andrés, JI; Alcázar, J; Alonso, JM; Alvarez, RM; Bakker, MH; Biesmans, I; Cid, JM; De Lucas, AI; Fernández, J; Font, LM; Hens, KA; Iturrino, L; Lenaerts, I; Martínez, S; Megens, AA; Pastor, J; Vermote, PC; Steckler, T Discovery of a new series of centrally active tricyclic isoxazoles combining serotonin (5-HT) reuptake inhibition with alpha2-adrenoceptor blocking activity. J Med Chem48:2054-71 (2005) [PubMed] Article
Andrés, JI; Alcázar, J; Alonso, JM; Alvarez, RM; Bakker, MH; Biesmans, I; Cid, JM; De Lucas, AI; Fernández, J; Font, LM; Hens, KA; Iturrino, L; Lenaerts, I; Martínez, S; Megens, AA; Pastor, J; Vermote, PC; Steckler, T Discovery of a new series of centrally active tricyclic isoxazoles combining serotonin (5-HT) reuptake inhibition with alpha2-adrenoceptor blocking activity. J Med Chem48:2054-71 (2005) [PubMed] Article