| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1 |
|---|
| Ligand | BDBM50330853 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_762068 (CHEMBL1816060) |
|---|
| IC50 | 24000±n/a nM |
|---|
| Citation |  Yamakawa, N; Suemasu, S; Matoyama, M; Tanaka, K; Katsu, T; Miyata, K; Okamoto, Y; Otsuka, M; Mizushima, T Synthesis and biological evaluation of loxoprofen derivatives. Bioorg Med Chem19:3299-311 (2011) [PubMed] Article Yamakawa, N; Suemasu, S; Matoyama, M; Tanaka, K; Katsu, T; Miyata, K; Okamoto, Y; Otsuka, M; Mizushima, T Synthesis and biological evaluation of loxoprofen derivatives. Bioorg Med Chem19:3299-311 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1 |
|---|
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | COX-1 | COX1 | Cyclooxygenase | Cyclooxygenase-1 | Cyclooxygenase-1 (COX-1) | PGH1_HUMAN | PTGS1 | Prostaglandin G/H synthase (cyclooxygenase) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68692.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P23219 |
|---|
| Residue: | 599 |
|---|
| Sequence: | MSRSLLLWFLLFLLLLPPLPVLLADPGAPTPVNPCCYYPCQHQGICVRFGLDRYQCDCTR
TGYSGPNCTIPGLWTWLRNSLRPSPSFTHFLLTHGRWFWEFVNATFIREMLMRLVLTVRS
NLIPSPPTYNSAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDAQLLARRF
LLRRKFIPDPQGTNLMFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNLERQ
YQLRLFKDGKLKYQVLDGEMYPPSVEEAPVLMHYPRGIPPQSQMAVGQEVFGLLPGLMLY
ATLWLREHNRVCDLLKAEHPTWGDEQLFQTTRLILIGETIKIVIEEYVQQLSGYFLQLKF
DPELLFGVQFQYRNRIAMEFNHLYHWHPLMPDSFKVGSQEYSYEQFLFNTSMLVDYGVEA
LVDAFSRQIAGRIGGGRNMDHHILHVAVDVIRESREMRLQPFNEYRKRFGMKPYTSFQEL
VGEKEMAAELEELYGDIDALEFYPGLLLEKCHPNSIFGESMIEIGAPFSLKGLLGNPICS
PEYWKPSTFGGEVGFNIVKTATLKKLVCLNTKTCPYVSFRVPDASQDDGPAVERPSTEL
|
|
|
|---|
| BDBM50330853 |
|---|
| n/a |
|---|
| Name | BDBM50330853 |
|---|
| Synonyms: | CHEMBL66552 | Sodium; 2-[4-(2-oxo-cyclopentylmethyl)-phenyl]-propionate; hydrate | loxoprofen sodium |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H17O3 |
|---|
| Mol. Mass. | 245.2942 |
|---|
| SMILES | CC(C([O-])=O)c1ccc(CC2CCCC2=O)cc1 |
|---|
| Structure | 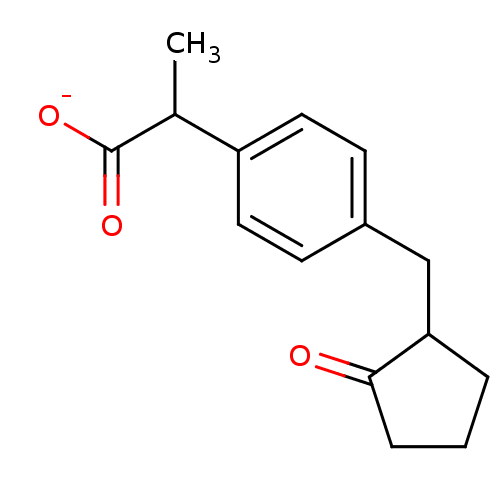
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yamakawa, N; Suemasu, S; Matoyama, M; Tanaka, K; Katsu, T; Miyata, K; Okamoto, Y; Otsuka, M; Mizushima, T Synthesis and biological evaluation of loxoprofen derivatives. Bioorg Med Chem19:3299-311 (2011) [PubMed] Article
Yamakawa, N; Suemasu, S; Matoyama, M; Tanaka, K; Katsu, T; Miyata, K; Okamoto, Y; Otsuka, M; Mizushima, T Synthesis and biological evaluation of loxoprofen derivatives. Bioorg Med Chem19:3299-311 (2011) [PubMed] Article