| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 5 |
|---|
| Ligand | BDBM50394586 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_857678 (CHEMBL2168514) |
|---|
| IC50 | 0.5±n/a nM |
|---|
| Citation |  Skerlj, R; Bridger, G; Zhou, Y; Bourque, E; McEachern, E; Danthi, S; Langille, J; Harwig, C; Veale, D; Carpenter, B; Ba, T; Bey, M; Baird, I; Wilson, T; Metz, M; MacFarland, R; Mosi, R; Bodart, V; Wong, R; Fricker, S; Huskens, D; Schols, D Mitigating hERG Inhibition: Design of Orally Bioavailable CCR5 Antagonists as Potent Inhibitors of R5 HIV-1 Replication. ACS Med Chem Lett3:216-221 (2012) [PubMed] Article Skerlj, R; Bridger, G; Zhou, Y; Bourque, E; McEachern, E; Danthi, S; Langille, J; Harwig, C; Veale, D; Carpenter, B; Ba, T; Bey, M; Baird, I; Wilson, T; Metz, M; MacFarland, R; Mosi, R; Bodart, V; Wong, R; Fricker, S; Huskens, D; Schols, D Mitigating hERG Inhibition: Design of Orally Bioavailable CCR5 Antagonists as Potent Inhibitors of R5 HIV-1 Replication. ACS Med Chem Lett3:216-221 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 5 |
|---|
| Name: | C-C chemokine receptor type 5 |
|---|
| Synonyms: | C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40540.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51681 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MDYQVSSPIYDINYYTSEPCQKINVKQIAARLLPPLYSLVFIFGFVGNMLVILILINCKR
LKSMTDIYLLNLAISDLFFLLTVPFWAHYAAAQWDFGNTMCQLLTGLYFIGFFSGIFFII
LLTIDRYLAVVHAVFALKARTVTFGVVTSVITWVVAVFASLPGIIFTRSQKEGLHYTCSS
HFPYSQYQFWKNFQTLKIVILGLVLPLLVMVICYSGILKTLLRCRNEKKRHRAVRLIFTI
MIVYFLFWAPYNIVLLLNTFQEFFGLNNCSSSNRLDQAMQVTETLGMTHCCINPIIYAFV
GEKFRNYLLVFFQKHIAKRFCKCCSIFQQEAPERASSVYTRSTGEQEISVGL
|
|
|
|---|
| BDBM50394586 |
|---|
| n/a |
|---|
| Name | BDBM50394586 |
|---|
| Synonyms: | CHEMBL2164212 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H38ClN5O4S |
|---|
| Mol. Mass. | 612.183 |
|---|
| SMILES | C[C@H](CCNC(=O)c1c(C)cc(Cl)nc1C)N1CCC(CC1)N(Cc1ccsc1)C(=O)NCc1ccc(cc1)C(O)=O |r| |
|---|
| Structure | 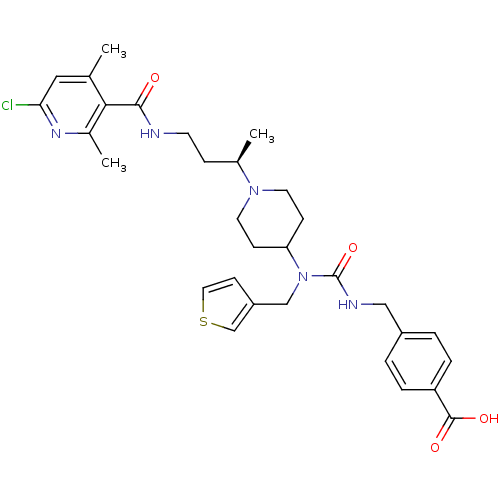
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Skerlj, R; Bridger, G; Zhou, Y; Bourque, E; McEachern, E; Danthi, S; Langille, J; Harwig, C; Veale, D; Carpenter, B; Ba, T; Bey, M; Baird, I; Wilson, T; Metz, M; MacFarland, R; Mosi, R; Bodart, V; Wong, R; Fricker, S; Huskens, D; Schols, D Mitigating hERG Inhibition: Design of Orally Bioavailable CCR5 Antagonists as Potent Inhibitors of R5 HIV-1 Replication. ACS Med Chem Lett3:216-221 (2012) [PubMed] Article
Skerlj, R; Bridger, G; Zhou, Y; Bourque, E; McEachern, E; Danthi, S; Langille, J; Harwig, C; Veale, D; Carpenter, B; Ba, T; Bey, M; Baird, I; Wilson, T; Metz, M; MacFarland, R; Mosi, R; Bodart, V; Wong, R; Fricker, S; Huskens, D; Schols, D Mitigating hERG Inhibition: Design of Orally Bioavailable CCR5 Antagonists as Potent Inhibitors of R5 HIV-1 Replication. ACS Med Chem Lett3:216-221 (2012) [PubMed] Article