| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytosolic phospholipase A2 |
|---|
| Ligand | BDBM50056714 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1454057 (CHEMBL3361803) |
|---|
| IC50 | 400±n/a nM |
|---|
| Citation |  Tomoo, T; Nakatsuka, T; Katayama, T; Hayashi, Y; Fujieda, Y; Terakawa, M; Nagahira, K Design, synthesis, and biological evaluation of 3-(1-Aryl-1H-indol-5-yl)propanoic acids as new indole-based cytosolic phospholipase A2a inhibitors. J Med Chem57:7244-62 (2014) [PubMed] Article Tomoo, T; Nakatsuka, T; Katayama, T; Hayashi, Y; Fujieda, Y; Terakawa, M; Nagahira, K Design, synthesis, and biological evaluation of 3-(1-Aryl-1H-indol-5-yl)propanoic acids as new indole-based cytosolic phospholipase A2a inhibitors. J Med Chem57:7244-62 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytosolic phospholipase A2 |
|---|
| Name: | Cytosolic phospholipase A2 |
|---|
| Synonyms: | CPLA2 | Lysophospholipase | PA24A_HUMAN | PLA2G4 | PLA2G4A | Phosphatidylcholine 2-acylhydrolase | Phospholipase A2 | Phospholipase A2 group IVA |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 85219.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P47712 |
|---|
| Residue: | 749 |
|---|
| Sequence: | MSFIDPYQHIIVEHQYSHKFTVVVLRATKVTKGAFGDMLDTPDPYVELFISTTPDSRKRT
RHFNNDINPVWNETFEFILDPNQENVLEITLMDANYVMDETLGTATFTVSSMKVGEKKEV
PFIFNQVTEMVLEMSLEVCSCPDLRFSMALCDQEKTFRQQRKEHIRESMKKLLGPKNSEG
LHSARDVPVVAILGSGGGFRAMVGFSGVMKALYESGILDCATYVAGLSGSTWYMSTLYSH
PDFPEKGPEEINEELMKNVSHNPLLLLTPQKVKRYVESLWKKKSSGQPVTFTDIFGMLIG
ETLIHNRMNTTLSSLKEKVNTAQCPLPLFTCLHVKPDVSELMFADWVEFSPYEIGMAKYG
TFMAPDLFGSKFFMGTVVKKYEENPLHFLMGVWGSAFSILFNRVLGVSGSQSRGSTMEEE
LENITTKHIVSNDSSDSDDESHEPKGTENEDAGSDYQSDNQASWIHRMIMALVSDSALFN
TREGRAGKVHNFMLGLNLNTSYPLSPLSDFATQDSFDDDELDAAVADPDEFERIYEPLDV
KSKKIHVVDSGLTFNLPYPLILRPQRGVDLIISFDFSARPSDSSPPFKELLLAEKWAKMN
KLPFPKIDPYVFDREGLKECYVFKPKNPDMEKDCPTIIHFVLANINFRKYRAPGVPRETE
EEKEIADFDIFDDPESPFSTFNFQYPNQAFKRLHDLMHFNTLNNIDVIKEAMVESIEYRR
QNPSRCSVSLSNVEARRFFNKEFLSKPKA
|
|
|
|---|
| BDBM50056714 |
|---|
| n/a |
|---|
| Name | BDBM50056714 |
|---|
| Synonyms: | CHEMBL3326964 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H34N2O5 |
|---|
| Mol. Mass. | 490.5907 |
|---|
| SMILES | NC(CO)(CO)CO.OC(=O)CCc1ccc2c(CCc3ccccc3)cn(-c3ccccc3)c2c1 |
|---|
| Structure | 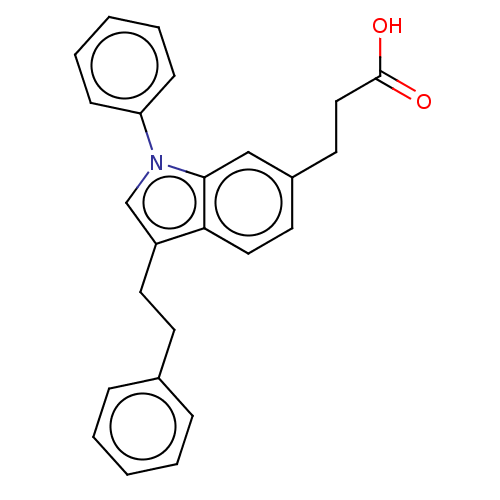
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tomoo, T; Nakatsuka, T; Katayama, T; Hayashi, Y; Fujieda, Y; Terakawa, M; Nagahira, K Design, synthesis, and biological evaluation of 3-(1-Aryl-1H-indol-5-yl)propanoic acids as new indole-based cytosolic phospholipase A2a inhibitors. J Med Chem57:7244-62 (2014) [PubMed] Article
Tomoo, T; Nakatsuka, T; Katayama, T; Hayashi, Y; Fujieda, Y; Terakawa, M; Nagahira, K Design, synthesis, and biological evaluation of 3-(1-Aryl-1H-indol-5-yl)propanoic acids as new indole-based cytosolic phospholipase A2a inhibitors. J Med Chem57:7244-62 (2014) [PubMed] Article