| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50158923 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Inhibition Assay (Testosterone Hydroxylase) |
|---|
| IC50 | 1.298e+4± 3253 nM |
|---|
| Citation |  Cashman, JR Synthetic compounds and derivatives as modulators of smoking or nicotine ingestion and lung cancer US Patent US8609708 Publication Date 12/17/2013 Cashman, JR Synthetic compounds and derivatives as modulators of smoking or nicotine ingestion and lung cancer US Patent US8609708 Publication Date 12/17/2013 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50158923 |
|---|
| n/a |
|---|
| Name | BDBM50158923 |
|---|
| Synonyms: | 5-Pyridin-3-yl-thiophene-2-carbaldehyde oxime | CHEMBL178534 | US8609708, 18 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C10H8N2OS |
|---|
| Mol. Mass. | 204.248 |
|---|
| SMILES | O=NCc1ccc(s1)-c1cccnc1 |
|---|
| Structure | 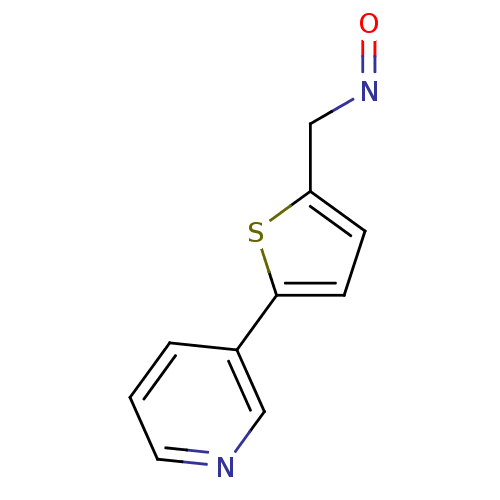
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cashman, JR Synthetic compounds and derivatives as modulators of smoking or nicotine ingestion and lung cancer US Patent US8609708 Publication Date 12/17/2013
Cashman, JR Synthetic compounds and derivatives as modulators of smoking or nicotine ingestion and lung cancer US Patent US8609708 Publication Date 12/17/2013