| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM508537 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | competition binding assay |
|---|
| Ki | 0.900±n/a nM |
|---|
| Citation |  Amjad, A; Gallo, GV; Henderson, TJ; Kuang, R; Lim, Y; Lo, MM; Metzger, E; de Lera Ruiz, M; Stamford, A; Tempest, P; Whitehead, B; Wu, H 2,2-difluorodioxolo A2A receptor antagonists US Patent US11046714 Publication Date 6/29/2021 Amjad, A; Gallo, GV; Henderson, TJ; Kuang, R; Lim, Y; Lo, MM; Metzger, E; de Lera Ruiz, M; Stamford, A; Tempest, P; Whitehead, B; Wu, H 2,2-difluorodioxolo A2A receptor antagonists US Patent US11046714 Publication Date 6/29/2021 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | A2A adenosine receptor (hA2A) | AA2AR_HUMAN | ADENOSINE A2 | ADENOSINE A2a | ADORA2 | ADORA2A | Adenosine A2A receptor (A2AAR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44716.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29274 |
|---|
| Residue: | 412 |
|---|
| Sequence: | MPIMGSSVYITVELAIAVLAILGNVLVCWAVWLNSNLQNVTNYFVVSLAAADIAVGVLAI
PFAITISTGFCAACHGCLFIACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGTR
AKGIIAICWVLSFAIGLTPMLGWNNCGQPKEGKNHSQGCGEGQVACLFEDVVPMNYMVYF
NFFACVLVPLLLMLGVYLRIFLAARRQLKQMESQPLPGERARSTLQKEVHAAKSLAIIVG
LFALCWLPLHIINCFTFFCPDCSHAPLWLMYLAIVLSHTNSVVNPFIYAYRIREFRQTFR
KIIRSHVLRQQEPFKAAGTSARVLAAHGSDGEQVSLRLNGHPPGVWANGSAPHPERRPNG
YALGLVSGGSAQESQGNTGLPDVELLSHELKGVCPEPPGLDDPLAQDGAGVS
|
|
|
|---|
| BDBM508537 |
|---|
| n/a |
|---|
| Name | BDBM508537 |
|---|
| Synonyms: | 8-[2-(2-cyclopropyl-6,7- dihydro[1,3]oxazolo[5,4- c]pyridin-5(4H)-yl)ethyl]-2,2- difluoro[1,3]dioxolo[4,5- h][1,2,4]triazolo[1,5- c]quinazolin-5-amine | US11046714, Example 14 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H19F2N7O3 |
|---|
| Mol. Mass. | 455.4175 |
|---|
| SMILES | Nc1nc2c3OC(F)(F)Oc3ccc2c2nc(CCN3CCc4nc(oc4C3)C3CC3)nn12 |
|---|
| Structure | 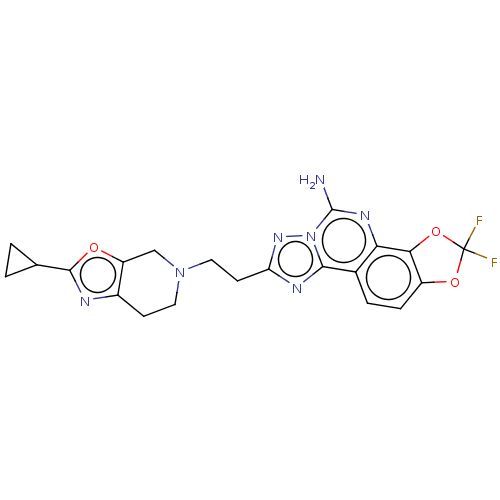
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Amjad, A; Gallo, GV; Henderson, TJ; Kuang, R; Lim, Y; Lo, MM; Metzger, E; de Lera Ruiz, M; Stamford, A; Tempest, P; Whitehead, B; Wu, H 2,2-difluorodioxolo A2A receptor antagonists US Patent US11046714 Publication Date 6/29/2021
Amjad, A; Gallo, GV; Henderson, TJ; Kuang, R; Lim, Y; Lo, MM; Metzger, E; de Lera Ruiz, M; Stamford, A; Tempest, P; Whitehead, B; Wu, H 2,2-difluorodioxolo A2A receptor antagonists US Patent US11046714 Publication Date 6/29/2021