| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Uromodulin |
|---|
| Ligand | BDBM534921 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | In-Vitro Assay |
|---|
| IC50 | 4.00±n/a nM |
|---|
| Citation |  Sharma, R; Iyer, P; Agarwal, S Substituted sulfoximine compounds US Patent US11236045 Publication Date 2/1/2022 Sharma, R; Iyer, P; Agarwal, S Substituted sulfoximine compounds US Patent US11236045 Publication Date 2/1/2022 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Uromodulin |
|---|
| Name: | Uromodulin |
|---|
| Synonyms: | THP | Tamm-Horsfall urinary glycoprotein | UMOD | UROM_HUMAN |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 69744.65 |
|---|
| Organism: | Human |
|---|
| Description: | P07911 |
|---|
| Residue: | 640 |
|---|
| Sequence: | MGQPSLTWMLMVVVASWFITTAATDTSEARWCSECHSNATCTEDEAVTTCTCQEGFTGDG
LTCVDLDECAIPGAHNCSANSSCVNTPGSFSCVCPEGFRLSPGLGCTDVDECAEPGLSHC
HALATCVNVVGSYLCVCPAGYRGDGWHCECSPGSCGPGLDCVPEGDALVCADPCQAHRTL
DEYWRSTEYGEGYACDTDLRGWYRFVGQGGARMAETCVPVLRCNTAAPMWLNGTHPSSDE
GIVSRKACAHWSGHCCLWDASVQVKACAGGYYVYNLTAPPECHLAYCTDPSSVEGTCEEC
SIDEDCKSNNGRWHCQCKQDFNITDISLLEHRLECGANDMKVSLGKCQLKSLGFDKVFMY
LSDSRCSGFNDRDNRDWVSVVTPARDGPCGTVLTRNETHATYSNTLYLADEIIIRDLNIK
INFACSYPLDMKVSLKTALQPMVSALNIRVGGTGMFTVRMALFQTPSYTQPYQGSSVTLS
TEAFLYVGTMLDGGDLSRFALLMTNCYATPSSNATDPLKYFIIQDRCPHTRDSTIQVVEN
GESSQGRFSVQMFRFAGNYDLVYLHCEVYLCDTMNEKCKPTCSGTRFRSGSVIDQSRVLN
LGPITRKGVQATVSRAFSSLGLLKVWLPLLLSATLTLTFQ
|
|
|
|---|
| BDBM534921 |
|---|
| n/a |
|---|
| Name | BDBM534921 |
|---|
| Synonyms: | N′-cyano-1-(4-cyanophenyl)-N-((1,2,3,5,6,7-hexahydro-s-indacen-4-yl)carbamoyl)-methanesulfonimidamide | US11236045, Example 66 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H21N5O2S |
|---|
| Mol. Mass. | 419.499 |
|---|
| SMILES | O=C(Nc1c2CCCc2cc2CCCc12)NS(=O)(Cc1ccc(cc1)C#N)=NC#N |
|---|
| Structure | 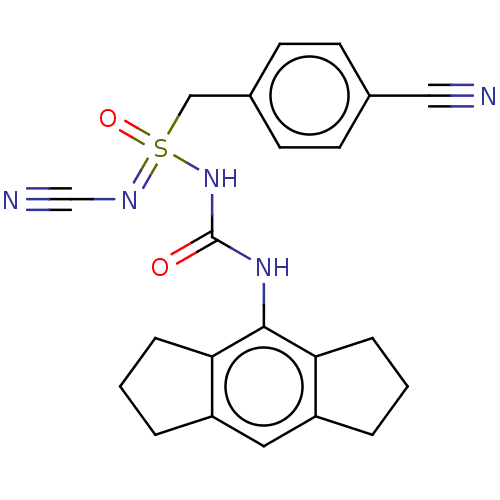
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sharma, R; Iyer, P; Agarwal, S Substituted sulfoximine compounds US Patent US11236045 Publication Date 2/1/2022
Sharma, R; Iyer, P; Agarwal, S Substituted sulfoximine compounds US Patent US11236045 Publication Date 2/1/2022