| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Dual specificity protein kinase TTK |
|---|
| Ligand | BDBM317473 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Radioactivity Based Kinase Assay |
|---|
| IC50 | 2.12±n/a nM |
|---|
| Citation |  Zhu, D; Boylan, J; Xu, S; Riggs, J; Shi, T; Wurmser, A; Mikolon, D; Deyanat-Yazdi, G Methods of treating a cancer using substituted pyrrolopyrimidine compounds, compositions thereof US Patent US9623028 Publication Date 4/18/2017 Zhu, D; Boylan, J; Xu, S; Riggs, J; Shi, T; Wurmser, A; Mikolon, D; Deyanat-Yazdi, G Methods of treating a cancer using substituted pyrrolopyrimidine compounds, compositions thereof US Patent US9623028 Publication Date 4/18/2017 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Dual specificity protein kinase TTK |
|---|
| Name: | Dual specificity protein kinase TTK |
|---|
| Synonyms: | Dual specificity protein kinase (TTK) | Dual specificity protein kinase TTK (MPS1) | MPS1 | MPS1L1 | Monopolar Spindle 1 (MPS1) | Monopolar Spindle 1 (Mps-1) | Monopolar Spindle 1 Kinase (MPS1) | Phosphotyrosine picked threonine-protein kinase (MPS1) | TTK | TTK_HUMAN |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 97086.79 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P33981 |
|---|
| Residue: | 857 |
|---|
| Sequence: | MESEDLSGRELTIDSIMNKVRDIKNKFKNEDLTDELSLNKISADTTDNSGTVNQIMMMAN
NPEDWLSLLLKLEKNSVPLSDALLNKLIGRYSQAIEALPPDKYGQNESFARIQVRFAELK
AIQEPDDARDYFQMARANCKKFAFVHISFAQFELSQGNVKKSKQLLQKAVERGAVPLEML
EIALRNLNLQKKQLLSEEEKKNLSASTVLTAQESFSGSLGHLQNRNNSCDSRGQTTKARF
LYGENMPPQDAEIGYRNSLRQTNKTKQSCPFGRVPVNLLNSPDCDVKTDDSVVPCFMKRQ
TSRSECRDLVVPGSKPSGNDSCELRNLKSVQNSHFKEPLVSDEKSSELIITDSITLKNKT
ESSLLAKLEETKEYQEPEVPESNQKQWQSKRKSECINQNPAASSNHWQIPELARKVNTEQ
KHTTFEQPVFSVSKQSPPISTSKWFDPKSICKTPSSNTLDDYMSCFRTPVVKNDFPPACQ
LSTPYGQPACFQQQQHQILATPLQNLQVLASSSANECISVKGRIYSILKQIGSGGSSKVF
QVLNEKKQIYAIKYVNLEEADNQTLDSYRNEIAYLNKLQQHSDKIIRLYDYEITDQYIYM
VMECGNIDLNSWLKKKKSIDPWERKSYWKNMLEAVHTIHQHGIVHSDLKPANFLIVDGML
KLIDFGIANQMQPDTTSVVKDSQVGTVNYMPPEAIKDMSSSRENGKSKSKISPKSDVWSL
GCILYYMTYGKTPFQQIINQISKLHAIIDPNHEIEFPDIPEKDLQDVLKCCLKRDPKQRI
SIPELLAHPYVQIQTHPVNQMAKGTTEEMKYVLGQLVGLNSPNSILKAAKTLYEHYSGGE
SHNSSSSKTFEKKRGKK
|
|
|
|---|
| BDBM317473 |
|---|
| n/a |
|---|
| Name | BDBM317473 |
|---|
| Synonyms: | 4-(5-(4-(1H-imidazol-2- yl)phenyl)-4-(tetrahydro-2H- pyran-4-yloxy)-7H-pyrrolo[2,3- d]pyrimidin-2-ylamino)-3- methoxy-N-methylbenzamide | US9623028, Compound 146 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H29N7O4 |
|---|
| Mol. Mass. | 539.5851 |
|---|
| SMILES | CNC(=O)c1ccc(Nc2nc(OC3CCOCC3)c3c(c[nH]c3n2)-c2ccc(cc2)-c2ncc[nH]2)c(OC)c1 |
|---|
| Structure | 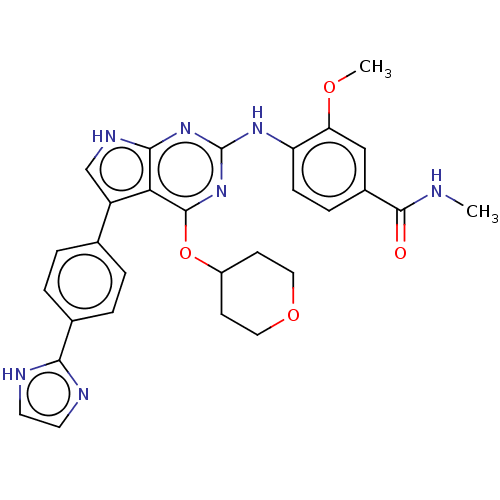
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhu, D; Boylan, J; Xu, S; Riggs, J; Shi, T; Wurmser, A; Mikolon, D; Deyanat-Yazdi, G Methods of treating a cancer using substituted pyrrolopyrimidine compounds, compositions thereof US Patent US9623028 Publication Date 4/18/2017
Zhu, D; Boylan, J; Xu, S; Riggs, J; Shi, T; Wurmser, A; Mikolon, D; Deyanat-Yazdi, G Methods of treating a cancer using substituted pyrrolopyrimidine compounds, compositions thereof US Patent US9623028 Publication Date 4/18/2017