| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bifunctional purine biosynthesis protein ATIC |
|---|
| Ligand | BDBM24685 |
|---|
| Substrate/Competitor | BDBM22579 |
|---|
| Meas. Tech. | AICAR Tfase Inhibition Assay |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 299.15±n/a K |
|---|
| Ki | >100000±n/a nM |
|---|
| Citation |  DeMartino, JK; Hwang, I; Connelly, S; Wilson, IA; Boger, DL Asymmetric synthesis of inhibitors of glycinamide ribonucleotide transformylase. J Med Chem51:5441-8 (2008) [PubMed] Article DeMartino, JK; Hwang, I; Connelly, S; Wilson, IA; Boger, DL Asymmetric synthesis of inhibitors of glycinamide ribonucleotide transformylase. J Med Chem51:5441-8 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Bifunctional purine biosynthesis protein ATIC |
|---|
| Name: | Bifunctional purine biosynthesis protein ATIC |
|---|
| Synonyms: | 5-aminoimidazole-4-carboxamide ribonucleotide formyltransferase | 5-aminoimidazole-4-carboxamide-ribonucleotide transformylase | AICAR Tfase | AICAR transformylase | ATIC | Aminoimidazole carboxamide ribonucleotide transformylase (AICAR Tfase) | Bifunctional purine biosynthesis protein PURH | IMP Cyclohydrolase (IMPCH) | IMP cyclohydrolase | IMP synthetase | Inosinicase | PUR9_HUMAN | PURH | Phosphoribosylaminoimidazolecarboxamide formyltransferase | Thymidylate synthase/GAR transformylase/AICAR transformylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 64616.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P31939 |
|---|
| Residue: | 592 |
|---|
| Sequence: | MAPGQLALFSVSDKTGLVEFARNLTALGLNLVASGGTAKALRDAGLAVRDVSELTGFPEM
LGGRVKTLHPAVHAGILARNIPEDNADMARLDFNLIRVVACNLYPFVKTVASPGVTVEEA
VEQIDIGGVTLLRAAAKNHARVTVVCEPEDYVVVSTEMQSSESKDTSLETRRQLALKAFT
HTAQYDEAISDYFRKQYSKGVSQMPLRYGMNPHQTPAQLYTLQPKLPITVLNGAPGFINL
CDALNAWQLVKELKEALGIPAAASFKHVSPAGAAVGIPLSEDEAKVCMVYDLYKTLTPIS
AAYARARGADRMSSFGDFVALSDVCDVPTAKIISREVSDGIIAPGYEEEALTILSKKKNG
NYCVLQMDQSYKPDENEVRTLFGLHLSQKRNNGVVDKSLFSNVVTKNKDLPESALRDLIV
ATIAVKYTQSNSVCYAKNGQVIGIGAGQQSRIHCTRLAGDKANYWWLRHHPQVLSMKFKT
GVKRAEISNAIDQYVTGTIGEDEDLIKWKALFEEVPELLTEAEKKEWVEKLTEVSISSDA
FFPFRDNVDRAKRSGVAYIAAPSGSAADKVVIEACDELGIILAHTNLRLFHH
|
|
|
|---|
| BDBM24685 |
|---|
| BDBM22579 |
|---|
| Name | BDBM24685 |
|---|
| Synonyms: | 10-thiomethyl-DDACTHF, 10R-3 | 10R (8) | 2-({4-[(1R)-4-(2,4-diamino-6-oxo-1,6-dihydropyrimidin-5-yl)-1-(methylsulfanyl)butyl]phenyl}formamido)pentanedioic acid |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H27N5O6S |
|---|
| Mol. Mass. | 477.534 |
|---|
| SMILES | CS[C@H](CCCc1c(N)nc(N)[nH]c1=O)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O |r| |
|---|
| Structure | 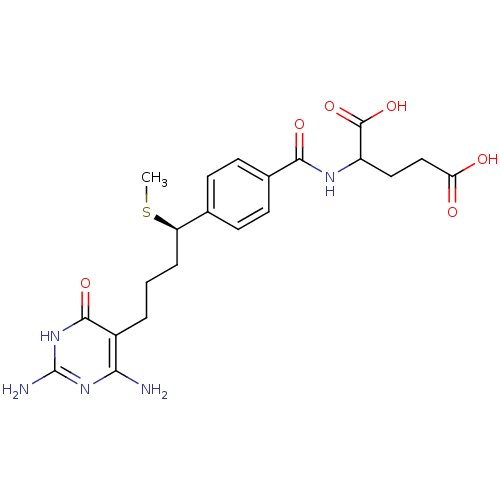
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 DeMartino, JK; Hwang, I; Connelly, S; Wilson, IA; Boger, DL Asymmetric synthesis of inhibitors of glycinamide ribonucleotide transformylase. J Med Chem51:5441-8 (2008) [PubMed] Article
DeMartino, JK; Hwang, I; Connelly, S; Wilson, IA; Boger, DL Asymmetric synthesis of inhibitors of glycinamide ribonucleotide transformylase. J Med Chem51:5441-8 (2008) [PubMed] Article