| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alkaline phosphatase, germ cell type |
|---|
| Ligand | BDBM39657 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Luminescent assay for HTS discovery of chemical inhibitors of placental alkaline phosphatase |
|---|
| IC50 | 999000±0 nM |
|---|
| Citation |  PubChem, PC Luminescent assay for HTS discovery of chemical inhibitors of placental alkaline phosphatase PubChem Bioassay(2007)[AID] PubChem, PC Luminescent assay for HTS discovery of chemical inhibitors of placental alkaline phosphatase PubChem Bioassay(2007)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Alkaline phosphatase, germ cell type |
|---|
| Name: | Alkaline phosphatase, germ cell type |
|---|
| Synonyms: | ALPG | ALPPL | ALPPL2 | Alkaline phosphatase placental-like | PPBN_HUMAN | placental-like alkaline phosphatase preproprotein |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 57374.31 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_29964 |
|---|
| Residue: | 532 |
|---|
| Sequence: | MQGPWVLLLLGLRLQLSLGIIPVEEENPDFWNRQAAEALGAAKKLQPAQTAAKNLIIFLG
DGMGVSTVTAARILKGQKKDKLGPETFLAMDRFPYVALSKTYSVDKHVPDSGATATAYLC
GVKGNFQTIGLSAAARFNQCNTTRGNEVISVMNRAKKAGKSVGVVTTTRVQHASPAGAYA
HTVNRNWYSDADVPASARQEGCQDIATQLISNMDIDVILGGGRKYMFPMGTPDPEYPDDY
SQGGTRLDGKNLVQEWLAKHQGARYVWNRTELLQASLDPSVTHLMGLFEPGDMKYEIHRD
STLDPSLMEMTEAALLLLSRNPRGFFLFVEGGRIDHGHHESRAYRALTETIMFDDAIERA
GQLTSEEDTLSLVTADHSHVFSFGGYPLRGSSIFGLAPGKARDRKAYTVLLYGNGPGYVL
KDGARPDVTESESGSPEYRQQSAVPLDGETHAGEDVAVFARGPQAHLVHGVQEQTFIAHV
MAFAACLEPYTACDLAPRAGTTDAAHPGPSVVPALLPLLAGTLLLLGTATAP
|
|
|
|---|
| BDBM39657 |
|---|
| n/a |
|---|
| Name | BDBM39657 |
|---|
| Synonyms: | MLS000523074 | N-(4-fluorobenzyl)-2-(2-keto-6-pyrrolidinosulfonyl-1,3-benzoxazol-3-yl)acetamide | N-[(4-fluorophenyl)methyl]-2-(2-oxidanylidene-6-pyrrolidin-1-ylsulfonyl-1,3-benzoxazol-3-yl)ethanamide | N-[(4-fluorophenyl)methyl]-2-(2-oxo-6-pyrrolidin-1-ylsulfonyl-1,3-benzoxazol-3-yl)acetamide | N-[(4-fluorophenyl)methyl]-2-[2-oxo-6-(1-pyrrolidinylsulfonyl)-1,3-benzoxazol-3-yl]acetamide | SMR000128339 | cid_9550167 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H20FN3O5S |
|---|
| Mol. Mass. | 433.453 |
|---|
| SMILES | Fc1ccc(CNC(=O)Cn2c3ccc(cc3oc2=O)S(=O)(=O)N2CCCC2)cc1 |
|---|
| Structure | 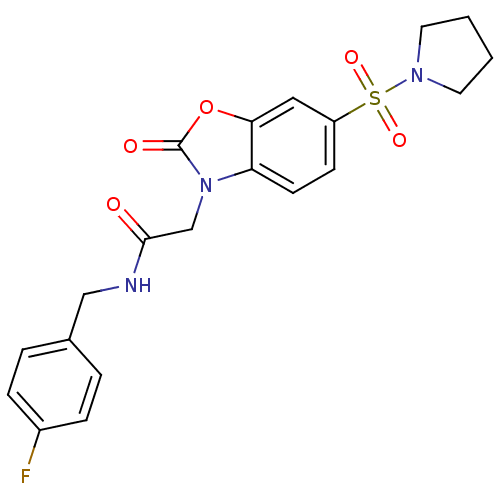
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Luminescent assay for HTS discovery of chemical inhibitors of placental alkaline phosphatase PubChem Bioassay(2007)[AID]
PubChem, PC Luminescent assay for HTS discovery of chemical inhibitors of placental alkaline phosphatase PubChem Bioassay(2007)[AID]