| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuropeptide Y receptor type 2 |
|---|
| Ligand | BDBM42421 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Dose response counterscreen assay for neuropeptide Y receptor Y1 (NPY-Y1): Cell-based high throughput assay to measure NPY-Y2 antagonism |
|---|
| IC50 | 30590±n/a nM |
|---|
| Citation |  PubChem, PC Dose response counterscreen assay for neuropeptide Y receptor Y1 (NPY-Y1): Cell-based high throughput assay to measure NPY-Y2 antagonism PubChem Bioassay(2008)[AID] PubChem, PC Dose response counterscreen assay for neuropeptide Y receptor Y1 (NPY-Y1): Cell-based high throughput assay to measure NPY-Y2 antagonism PubChem Bioassay(2008)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Neuropeptide Y receptor type 2 |
|---|
| Name: | Neuropeptide Y receptor type 2 |
|---|
| Synonyms: | NPY-Y2 | NPY-Y2 receptor | NPY2-R | NPY2R | NPY2R_HUMAN | Neuropeptide Y receptor type 2 | Y2 receptor | neuropeptide Y receptor Y2 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 42734.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | NPY-Y2 NPY2R HUMAN::P49146 |
|---|
| Residue: | 381 |
|---|
| Sequence: | MGPIGAEADENQTVEEMKVEQYGPQTTPRGELVPDPEPELIDSTKLIEVQVVLILAYCSI
ILLGVIGNSLVIHVVIKFKSMRTVTNFFIANLAVADLLVNTLCLPFTLTYTLMGEWKMGP
VLCHLVPYAQGLAVQVSTITLTVIALDRHRCIVYHLESKISKRISFLIIGLAWGISALLA
SPLAIFREYSLIEIIPDFEIVACTEKWPGEEKSIYGTVYSLSSLLILYVLPLGIISFSYT
RIWSKLKNHVSPGAANDHYHQRRQKTTKMLVCVVVVFAVSWLPLHAFQLAVDIDSQVLDL
KEYKLIFTVFHIIAMCSTFANPLLYGWMNSNYRKAFLSAFRCEQRLDAIHSEVSVTFKAK
KNLEVRKNSGPNDSFTEATNV
|
|
|
|---|
| BDBM42421 |
|---|
| n/a |
|---|
| Name | BDBM42421 |
|---|
| Synonyms: | 2-[[[2-(4-chlorophenyl)-5-methyl-3-pyrazolyl]amino]methylidene]propanedioic acid diethyl ester | 2-[[[2-(4-chlorophenyl)-5-methyl-pyrazol-3-yl]amino]methylene]malonic acid diethyl ester | MLS000541363 | SMR000126221 | cid_3849628 | diethyl 2-({[1-(4-chlorophenyl)-3-methyl-1H-pyrazol-5-yl]amino}methylene)malonate | diethyl 2-[[[2-(4-chlorophenyl)-5-methyl-pyrazol-3-yl]amino]methylidene]propanedioate | diethyl 2-[[[2-(4-chlorophenyl)-5-methylpyrazol-3-yl]amino]methylidene]propanedioate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H20ClN3O4 |
|---|
| Mol. Mass. | 377.822 |
|---|
| SMILES | CCOC(=O)C(C=Nc1cc(C)nn1-c1ccc(Cl)cc1)C(=O)OCC |w:7.7| |
|---|
| Structure | 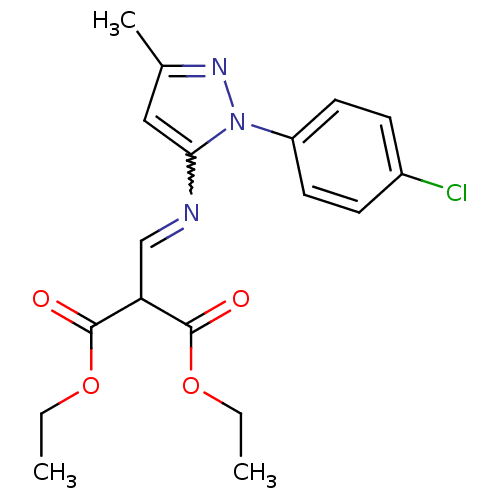
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Dose response counterscreen assay for neuropeptide Y receptor Y1 (NPY-Y1): Cell-based high throughput assay to measure NPY-Y2 antagonism PubChem Bioassay(2008)[AID]
PubChem, PC Dose response counterscreen assay for neuropeptide Y receptor Y1 (NPY-Y1): Cell-based high throughput assay to measure NPY-Y2 antagonism PubChem Bioassay(2008)[AID]