| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glutathione S-transferase Mu 1 |
|---|
| Ligand | BDBM44483 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Profiling Assay to determine GST-GSH interactions in multiplex bead-based assays |
|---|
| EC50 | 201.5±n/a nM |
|---|
| Citation |  PubChem, PC Profiling Assay to determine GST-GSH interactions in multiplex bead-based assays PubChem Bioassay(2010)[AID] PubChem, PC Profiling Assay to determine GST-GSH interactions in multiplex bead-based assays PubChem Bioassay(2010)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Glutathione S-transferase Mu 1 |
|---|
| Name: | Glutathione S-transferase Mu 1 |
|---|
| Synonyms: | GST class-mu 1 | GST1 | GSTM1 | GSTM1-1 | GSTM1_HUMAN | GSTM1a-1a | GSTM1b-1b | GTH4 | Glutathione S-transferase Mu 1 | HB subunit 4 | glutathione S-transferase |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 25712.03 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_799596 |
|---|
| Residue: | 218 |
|---|
| Sequence: | MPMILGYWDIRGLAHAIRLLLEYTDSSYEEKKYTMGDAPDYDRSQWLNEKFKLGLDFPNL
PYLIDGAHKITQSNAILCYIARKHNLCGETEEEKIRVDILENQTMDNHMQLGMICYNPEF
EKLKPKYLEELPEKLKLYSEFLGKRPWFAGNKITFVDFLVYDVLDLHRIFEPKCLDAFPN
LKDFISRFEGLEKISAYMKSSRFLPRPVFSKMAVWGNK
|
|
|
|---|
| BDBM44483 |
|---|
| n/a |
|---|
| Name | BDBM44483 |
|---|
| Synonyms: | (5Z)-3-butan-2-yl-5-[[9-methyl-4-oxidanylidene-2-(3-propan-2-yloxypropylamino)pyrido[1,2-a]pyrimidin-3-yl]methylidene]-2-sulfanylidene-1,3-thiazolidin-4-one | (5Z)-3-butan-2-yl-5-[[9-methyl-4-oxo-2-(3-propan-2-yloxypropylamino)-3-pyrido[1,2-a]pyrimidinyl]methylidene]-2-sulfanylidene-4-thiazolidinone | (5Z)-3-butan-2-yl-5-[[9-methyl-4-oxo-2-(3-propan-2-yloxypropylamino)pyrido[1,2-a]pyrimidin-3-yl]methylidene]-2-sulfanylidene-1,3-thiazolidin-4-one | (5Z)-5-[[2-(3-isopropoxypropylamino)-4-keto-9-methyl-pyrido[1,2-a]pyrimidin-3-yl]methylene]-3-sec-butyl-2-thioxo-thiazolidin-4-one | 3-[3-sec-Butyl-4-oxo-2-thioxo-thiazolidin-(5Z)-ylidenemethyl]-2-(3-isopropoxy-propylamino)-9-methyl-py rido[1,2-a]pyrimidin-4-one | MLS000557019 | SMR000148136 | cid_6297320 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H30N4O3S2 |
|---|
| Mol. Mass. | 474.639 |
|---|
| SMILES | CCC(C)N1C(=S)S\C(=C/c2c(NCCCOC(C)C)nc3c(C)cccn3c2=O)C1=O |
|---|
| Structure | 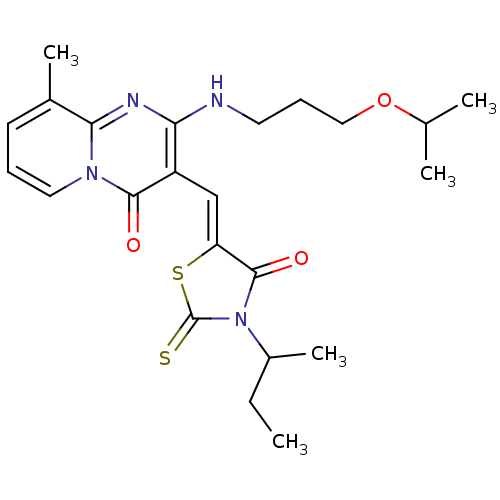
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Profiling Assay to determine GST-GSH interactions in multiplex bead-based assays PubChem Bioassay(2010)[AID]
PubChem, PC Profiling Assay to determine GST-GSH interactions in multiplex bead-based assays PubChem Bioassay(2010)[AID]