| Reaction Details |
|---|
 | Report a problem with these data |
| Target | SUMO-activating enzyme subunit 1 |
|---|
| Ligand | BDBM61211 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | uHTS HTRF assay for identification of inhibitors of SUMOylation |
|---|
| IC50 | 7540±n/a nM |
|---|
| Citation |  PubChem, PC uHTS HTRF assay for identification of inhibitors of SUMOylation PubChem Bioassay(2010)[AID] PubChem, PC uHTS HTRF assay for identification of inhibitors of SUMOylation PubChem Bioassay(2010)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| SUMO-activating enzyme subunit 1 |
|---|
| Name: | SUMO-activating enzyme subunit 1 |
|---|
| Synonyms: | AOS1 | SAE1 | SAE1_HUMAN | SUA1 | SUMO-activating enzyme subunit 1 (SAE1) | SUMO1 activating enzyme subunit 1 | UBLE1A |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 38440.84 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_17390638 |
|---|
| Residue: | 346 |
|---|
| Sequence: | MVEKEEAGGGISEEEAAQYDRQIRLWGLEAQKRLRASRVLLVGLKGLGAEIAKNLILAGV
KGLTMLDHEQVTPEDPGAQFLIRTGSVGRNRAEASLERAQNLNPMVDVKVDTEDIEKKPE
SFFTQFDAVCLTCCSRDVIVKVDQICHKNSIKFFTGDVFGYHGYTFANLGEHEFVEEKTK
VAKVSQGVEDGPDTKRAKLDSSETTMVKKKVVFCPVKEALEVDWSSEKAKAALKRTTSDY
FLLQVLLKFRTDKGRDPSSDTYEEDSELLLQIRNDVLDSLGISPDLLPEDFVRYCFSEMA
PVCAVVGGILAQEIVKALSQRDPPHNNFFFFDGMKGNGIVECLGPK
|
|
|
|---|
| BDBM61211 |
|---|
| n/a |
|---|
| Name | BDBM61211 |
|---|
| Synonyms: | (6E)-5-azanylidene-6-[[2,5-dimethyl-1-(phenylmethyl)pyrrol-3-yl]methylidene]-2-(furan-2-yl)-[1,3,4]thiadiazolo[3,2-a]pyrimidin-7-one | (6E)-6-[(1-benzyl-2,5-dimethyl-pyrrol-3-yl)methylene]-2-(2-furyl)-5-imino-[1,3,4]thiadiazolo[3,2-a]pyrimidin-7-one | (6E)-6-[(1-benzyl-2,5-dimethylpyrrol-3-yl)methylidene]-2-(furan-2-yl)-5-imino-[1,3,4]thiadiazolo[3,2-a]pyrimidin-7-one | (6E)-6-[[2,5-dimethyl-1-(phenylmethyl)-3-pyrrolyl]methylidene]-2-(2-furanyl)-5-imino-[1,3,4]thiadiazolo[3,2-a]pyrimidin-7-one | 6-[1-(1-Benzyl-2,5-dimethyl-1H-pyrrol-3-yl)-meth-(E)-ylidene]-2-furan-2-yl-5-imino-5,6-dihydro-[1,3,4]thia diazolo[3,2-a]pyrimidin-7-one | MLS000762668 | SMR000438147 | cid_16422746 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H19N5O2S |
|---|
| Mol. Mass. | 429.494 |
|---|
| SMILES | Cc1cc(\C=C2\C(=O)N=C3SC(=NN3C2=N)c2ccco2)c(C)n1Cc1ccccc1 |c:11,t:8| |
|---|
| Structure | 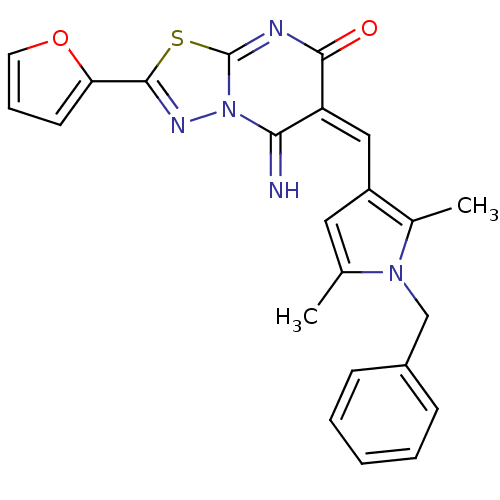
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC uHTS HTRF assay for identification of inhibitors of SUMOylation PubChem Bioassay(2010)[AID]
PubChem, PC uHTS HTRF assay for identification of inhibitors of SUMOylation PubChem Bioassay(2010)[AID]