| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Vacuolar aminopeptidase 1 |
|---|
| Ligand | BDBM50142887 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Dose Response of TOR pathway GFP-fusion proteins in Saccharomyes cerevisiae specifically LAP4 based on MLPCN hits |
|---|
| EC50 | 4823±n/a nM |
|---|
| Citation |  PubChem, PC Dose Response of TOR pathway GFP-fusion proteins in Saccharomyes cerevisiae specifically LAP4 based on MLPCN hits PubChem Bioassay(2010)[AID] PubChem, PC Dose Response of TOR pathway GFP-fusion proteins in Saccharomyes cerevisiae specifically LAP4 based on MLPCN hits PubChem Bioassay(2010)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Vacuolar aminopeptidase 1 |
|---|
| Name: | Vacuolar aminopeptidase 1 |
|---|
| Synonyms: | AMPL_YEAST | APE1 | API | LAP4 | YSC1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 57084.90 |
|---|
| Organism: | Saccharomyces cerevisiae |
|---|
| Description: | gi_486173 |
|---|
| Residue: | 514 |
|---|
| Sequence: | MEEQREILEQLKKTLQMLTVEPSKNNQIANEEKEKKENENSWCILEHNYEDIAQEFIDFI
YKNPTTYHVVSFFAELLDKHNFKYLSEKSNWQDSIGEDGGKFYTIRNGTNLSAFILGKNW
RAEKGVGVIGSHVDALTVKLKPVSFKDTAEGYGRIAVAPYGGTLNELWLDRDLGIGGRLL
YKKKGTNEIKSALVDSTPLPVCRIPSLAPHFGKPAEGPFDKEDQTIPVIGFPTPDEEGNE
PPTDDEKKSPLFGKHCIHLLRYVAKLAGVEVSELIQMDLDLFDVQKGTIGGIGKHFLFAP
RLDDRLCSFAAMIALICYAKDVNTEESDLFSTVTLYDNEEIGSLTRQGAKGGLLESVVER
SSSAFTKKPVDLHTVWANSIILSADVNHLYNPNFPEVYLKNHFPVPNVGITLSLDPNGHM
ATDVVGTALVEELARRNGDKVQYFQIKNNSRSGGTIGPSLASQTGARTIDLGIAQLSMHS
IRAATGSKDVGLGVKFFNGFFKHWRSVYDEFGEL
|
|
|
|---|
| BDBM50142887 |
|---|
| n/a |
|---|
| Name | BDBM50142887 |
|---|
| Synonyms: | 1-(tert-butyl)-3-(4-chlorophenyl)-4-aminopyrazolo[3,4-d]pyrimidine | 1-tert-Butyl-3-(4-chloro-phenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-ylamine | 1-tert-butyl-3-(4-chlorophenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine | 4-Amino-5-(4-chlorophenyl)-7-(t-butyl)pyrazolo[3,4-d]pyrimidine | 4-amino-5-(4-chlorophenyl)-7-(tert-butyl)pyrazolo[3,4-d]pyrimidine | AG-1879 | CHEMBL406845 | PP2 | US10544104, Compound 146 | US10688093, Compound PP2 (Pfizer) | US11701353, PP2 (Pfizer) | US9765037, Compound 146 | cid_4878 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H16ClN5 |
|---|
| Mol. Mass. | 301.774 |
|---|
| SMILES | CC(C)(C)n1nc(-c2ccc(Cl)cc2)c2c(N)ncnc12 |
|---|
| Structure | 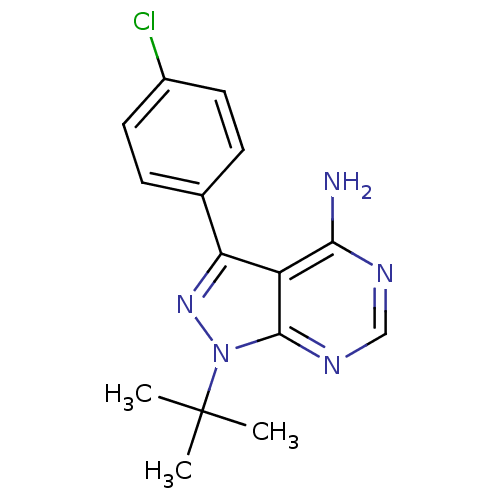
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Dose Response of TOR pathway GFP-fusion proteins in Saccharomyes cerevisiae specifically LAP4 based on MLPCN hits PubChem Bioassay(2010)[AID]
PubChem, PC Dose Response of TOR pathway GFP-fusion proteins in Saccharomyes cerevisiae specifically LAP4 based on MLPCN hits PubChem Bioassay(2010)[AID]