| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Beta-arrestin-1 |
|---|
| Ligand | BDBM50004656 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | >10000±n/a nM |
|---|
| Comments | PDSP_467 |
|---|
| Citation |  Bloom, JW; Halonen, M; Lawrence, LJ; Rould, E; Seaver, NA; Yamamura, HI Characterization of high affinity [3H]pirenzepine and (-)-[3H] quinuclidinyl benzilate binding to muscarinic cholinergic receptors in rabbit peripheral lung. J Pharmacol Exp Ther240:51-8 (1987) [PubMed] Bloom, JW; Halonen, M; Lawrence, LJ; Rould, E; Seaver, NA; Yamamura, HI Characterization of high affinity [3H]pirenzepine and (-)-[3H] quinuclidinyl benzilate binding to muscarinic cholinergic receptors in rabbit peripheral lung. J Pharmacol Exp Ther240:51-8 (1987) [PubMed] |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Beta-arrestin-1 |
|---|
| Name: | Beta-arrestin-1 |
|---|
| Synonyms: | ARRB1 | ARRB1_RABIT | Beta-arrestin-1 | Cholinergic, muscarinic | OPIATE Delta | PAF Platelet activating factor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 46360.54 |
|---|
| Organism: | RABBIT |
|---|
| Description: | Cholinergic, muscarinic 0 RABBIT::Q95223 |
|---|
| Residue: | 410 |
|---|
| Sequence: | MGDKGTRVFKKASPNGKLTVYLGKRGFVDHIDLVDPVDGVVLVDPEYLKERRVYVTLTCA
FRYGREDLDVLGLTFRKDLFVANVQSFPPAPEDKKPLTRLQERLIKKLGEHAYPFTFEIP
PKLPCSVTLQPGPEDTGKACGVDYEVKAFCAENLEEKIHKRNSVRLVIRKVQYAPERPGP
HPTAETTRLFLMSDKPLHLEASLDKEIYYHGEPIIVNVHVTNNTNKTVKKIKISVRQYAD
ICLFNTAQYKCPVAMEEADDTVAPSSTFCKVYTLTPFLANNREKRGLALDGKLKHEDTNL
ASSTLMREGANREILGIIVSYKVKVKLVVSRGGDVAVELPFTLMHPKPKEEPPHREVPEN
ETPVDTNLIELDTNDDDIVFEDFARQRLKGMKDDKEEEDDVTGSPRLNDR
|
|
|
|---|
| BDBM50004656 |
|---|
| n/a |
|---|
| Name | BDBM50004656 |
|---|
| Synonyms: | (2-Carbamoyloxy-ethyl)-trimethyl-ammonium | (2-Carbamoyloxyethyl)trimethylammonium chloride | (2-Hydroxyethyl)trimethyl ammonium chloride carbamate | (2-Hydroxyethyl)trimethylammonium chloride carbamate | (carbachol)(2-Carbamoyloxy-ethyl)-trimethyl-ammonium | 2-((Aminocarbonyl)oxy)-N,N,N-trimethylethanaminium chloride | 2-((Aminocarbonyl)oxy)-N,N,N-trimethylethanaminum chloride | 2-(carbamoyloxy)-N,N,N-trimethylethanaminium | 2-(carbamoyloxy)-N,N,N-trimethylethanaminium chloride | CARBACHOL | CARBASTAT | CHEMBL14 | Carbachol chloride | Carbamylcholine | Choline carbamate chloride | Choline chloride, carbamate | Choline chlorine carbamate | Karbachol | Karbamoylcholin chlorid | MIOSTAT |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C6H15N2O2 |
|---|
| Mol. Mass. | 147.195 |
|---|
| SMILES | C[N+](C)(C)CCOC(N)=O |
|---|
| Structure | 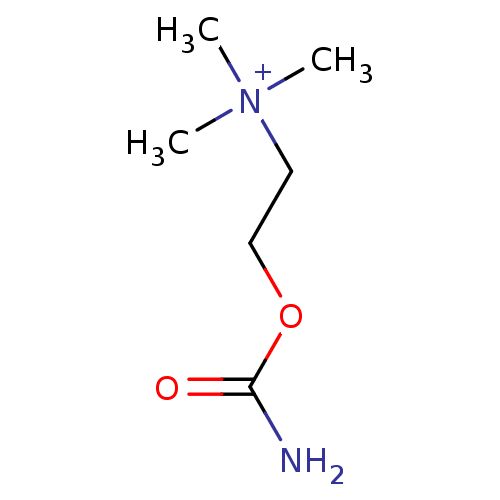
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bloom, JW; Halonen, M; Lawrence, LJ; Rould, E; Seaver, NA; Yamamura, HI Characterization of high affinity [3H]pirenzepine and (-)-[3H] quinuclidinyl benzilate binding to muscarinic cholinergic receptors in rabbit peripheral lung. J Pharmacol Exp Ther240:51-8 (1987) [PubMed]
Bloom, JW; Halonen, M; Lawrence, LJ; Rould, E; Seaver, NA; Yamamura, HI Characterization of high affinity [3H]pirenzepine and (-)-[3H] quinuclidinyl benzilate binding to muscarinic cholinergic receptors in rabbit peripheral lung. J Pharmacol Exp Ther240:51-8 (1987) [PubMed]