| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholecystokinin |
|---|
| Ligand | BDBM50019260 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | 2.1±n/a nM |
|---|
| Comments | PDSP_45 |
|---|
| Citation |  Evans, BE; Bock, MG; Rittle, KE; DiPardo, RM; Whitter, WL; Veber, DF; Anderson, PS; Freidinger, RM Design of potent, orally effective, nonpeptidal antagonists of the peptide hormone cholecystokinin. Proc Natl Acad Sci U S A83:4918-22 (1986) [PubMed] Article Evans, BE; Bock, MG; Rittle, KE; DiPardo, RM; Whitter, WL; Veber, DF; Anderson, PS; Freidinger, RM Design of potent, orally effective, nonpeptidal antagonists of the peptide hormone cholecystokinin. Proc Natl Acad Sci U S A83:4918-22 (1986) [PubMed] Article |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Cholecystokinin |
|---|
| Name: | Cholecystokinin |
|---|
| Synonyms: | CCKN_RAT | Cck |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 12844.29 |
|---|
| Organism: | RAT |
|---|
| Description: | Cholecystokinin 0 RAT::P01355 |
|---|
| Residue: | 115 |
|---|
| Sequence: | MKCGVCLCVVMAVLAAGALAQPVVPVEAVDPMEQRAEEAPRRQLRAVLRPDSEPRARLGA
LLARYIQQVRKAPSGRMSVLKNLQGLDPSHRISDRDYMGWMDFGRRSAEDYEYPS
|
|
|
|---|
| BDBM50019260 |
|---|
| n/a |
|---|
| Name | BDBM50019260 |
|---|
| Synonyms: | 1H-Indole-2-carboxylic acid [5-(2-fluoro-phenyl)-2-oxo-2,3-dihydro-1H-benzo[e][1,4]diazepin-3-yl]-amide | 1H-Indole-2-carboxylic acid [5-(2-fluoro-phenyl)-2-oxo-2,3-dihydro-1H-benzo[e][1,4]diazepin-3-yl]-amide: 0.16CH2Cl2 | CCK antagonist synthetic 21 | CHEMBL306806 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H17FN4O2 |
|---|
| Mol. Mass. | 412.4158 |
|---|
| SMILES | Fc1ccccc1C1=NC(NC(=O)c2cc3ccccc3[nH]2)C(=O)Nc2ccccc12 |t:8| |
|---|
| Structure | 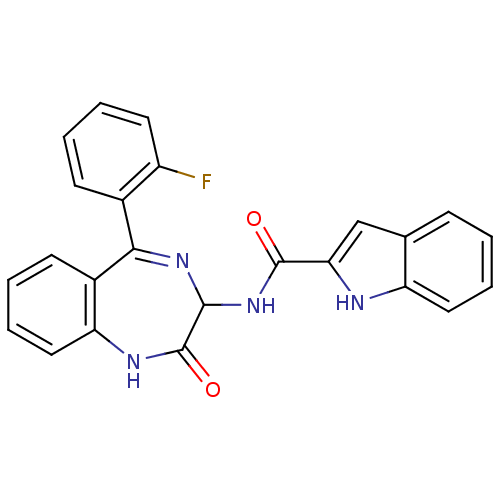
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Evans, BE; Bock, MG; Rittle, KE; DiPardo, RM; Whitter, WL; Veber, DF; Anderson, PS; Freidinger, RM Design of potent, orally effective, nonpeptidal antagonists of the peptide hormone cholecystokinin. Proc Natl Acad Sci U S A83:4918-22 (1986) [PubMed] Article
Evans, BE; Bock, MG; Rittle, KE; DiPardo, RM; Whitter, WL; Veber, DF; Anderson, PS; Freidinger, RM Design of potent, orally effective, nonpeptidal antagonists of the peptide hormone cholecystokinin. Proc Natl Acad Sci U S A83:4918-22 (1986) [PubMed] Article