

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Type-1 angiotensin II receptor A | ||
| Ligand | BDBM50009338 | ||
| Substrate/Competitor | n/a | ||
| Ki | 1.7±n/a nM | ||
| Comments | PDSP_1661 | ||
| Citation |  Keiser, JA; Ryan, MJ; Panek, RL; Hodges, JC; Sircar, I Pharmacologic characterization of CI-996, a new angiotensin receptor antagonist. J Pharmacol Exp Ther272:963-9 (1995) [PubMed] Keiser, JA; Ryan, MJ; Panek, RL; Hodges, JC; Sircar, I Pharmacologic characterization of CI-996, a new angiotensin receptor antagonist. J Pharmacol Exp Ther272:963-9 (1995) [PubMed] | ||
| More Info.: | Get all data from this article | ||
| Type-1 angiotensin II receptor A | |||
| Name: | Type-1 angiotensin II receptor A | ||
| Synonyms: | AGTRA_RAT | ANGIOTENSIN AT1 | Agtr1 | Agtr1a | Angiotensin II AT1 | Angiotensin II AT1A | Angiotensin II receptor (AT-1) type-1 | At1a | Type-1A angiotensin II receptor | ||
| Type: | Enzyme Catalytic Domain | ||
| Mol. Mass.: | 40910.53 | ||
| Organism: | RAT | ||
| Description: | ANGIOTENSIN AT1 AGTR1 RAT::P25095 | ||
| Residue: | 359 | ||
| Sequence: |
| ||
| BDBM50009338 | |||
| n/a | |||
| Name | BDBM50009338 | ||
| Synonyms: | (S)-2-((S)-1-((S)-2-((S)-2-((S)-2-((S)-2-((S)-5-(diaminomethyleneamino)-2-(2-(methylamino)acetamido)pentanamido)-3-methylbutanamido)-3-(4-hydroxyphenyl)propanamido)-3-methylbutanamido)-3-(1H-imidazol-5-yl)propanoyl)pyrrolidine-5-carboxamido)propanoic acid | CHEMBL938 | SARALASIN | Sar-Arg-Val-Tyr-Val-His-Pro-Ala | Sar-Arg-Val-Tyr-Val-His-Pro-Ala (sarlasin) | [Sar1-Arg2-Val3-Tyr4-Val5-His6-Pro7-Ala8] | ||
| Type | Small organic molecule | ||
| Emp. Form. | C42H65N13O10 | ||
| Mol. Mass. | 912.0466 | ||
| SMILES | CNCC(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(O)=O |r,wU:60.63,wD:24.23,36.36,43.43,56.59,17.16,6.5,(6.84,-31.4,;5.3,-31.4,;4.53,-30.07,;2.99,-30.07,;2.22,-31.4,;2.22,-28.73,;2.99,-27.4,;4.53,-27.4,;5.3,-26.07,;4.53,-24.73,;5.3,-23.4,;6.84,-23.4,;7.61,-24.73,;7.61,-22.07,;2.22,-26.07,;2.99,-24.73,;.68,-26.07,;-.09,-24.73,;.68,-23.4,;2.22,-23.4,;-.09,-22.07,;-1.63,-24.73,;-2.4,-23.4,;-2.4,-26.07,;-3.94,-26.07,;-4.71,-24.73,;-6.25,-24.73,;-7.02,-23.4,;-8.56,-23.4,;-9.33,-24.73,;-10.87,-24.73,;-8.56,-26.07,;-7.02,-26.07,;-4.71,-27.4,;-6.25,-27.4,;-3.94,-28.73,;-4.71,-30.07,;-6.25,-30.07,;-7.02,-28.73,;-7.02,-31.4,;-3.94,-31.4,;-2.4,-31.4,;-4.71,-32.73,;-3.94,-34.07,;-4.71,-35.4,;-6.25,-35.4,;-7.15,-34.16,;-8.62,-34.63,;-8.62,-36.17,;-7.15,-36.65,;-2.4,-34.07,;-1.63,-32.73,;-1.63,-35.4,;-2.26,-36.81,;-1.12,-37.84,;.22,-37.07,;-.1,-35.56,;.93,-34.42,;.45,-32.95,;2.42,-34.82,;3.5,-33.73,;3.11,-32.24,;5,-34.13,;6.09,-33.04,;5.39,-35.61,)| | ||
| Structure | 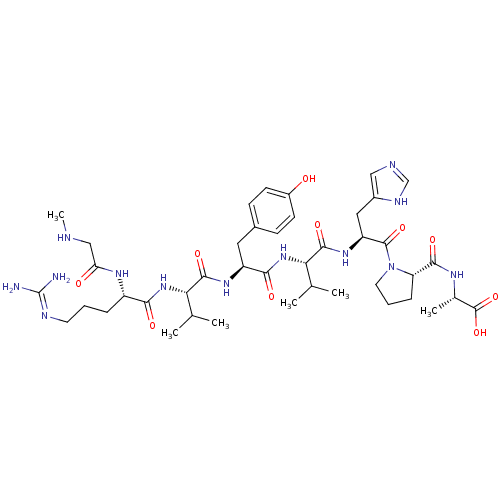
| ||