

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Gastrin/cholecystokinin type B receptor | ||
| Ligand | BDBM82403 | ||
| Substrate/Competitor | n/a | ||
| Ki | 6.3±n/a nM | ||
| Comments | PDSP_2204 | ||
| Citation |  Nishida, A; Miyata, K; Tsutsumi, R; Yuki, H; Akuzawa, S; Kobayashi, A; Kamato, T; Ito, H; Yamano, M; Katuyama, Y Pharmacological profile of (R)-1-[2,3-dihydro-1-(2'-methylphenacyl)-2-oxo- 5-phenyl-1H-1,4-benzodiazepin-3-yl]-3-(3-methylphenyl)urea (YM022), a new potent and selective gastrin/cholecystokinin-B receptor antagonist, in vitro and in vivo. J Pharmacol Exp Ther269:725-31 (1994) [PubMed] Nishida, A; Miyata, K; Tsutsumi, R; Yuki, H; Akuzawa, S; Kobayashi, A; Kamato, T; Ito, H; Yamano, M; Katuyama, Y Pharmacological profile of (R)-1-[2,3-dihydro-1-(2'-methylphenacyl)-2-oxo- 5-phenyl-1H-1,4-benzodiazepin-3-yl]-3-(3-methylphenyl)urea (YM022), a new potent and selective gastrin/cholecystokinin-B receptor antagonist, in vitro and in vivo. J Pharmacol Exp Ther269:725-31 (1994) [PubMed] | ||
| More Info.: | Get all data from this article | ||
| Gastrin/cholecystokinin type B receptor | |||
| Name: | Gastrin/cholecystokinin type B receptor | ||
| Synonyms: | Cckbr | Cholecystokinin A | Cholecystokinin B receptor | Cholecystokinin receptor | GASR_RAT | Gastrin/cholecystokinin type B receptor | ||
| Type: | Enzyme Catalytic Domain | ||
| Mol. Mass.: | 48980.43 | ||
| Organism: | RAT | ||
| Description: | Cholecystokinin A CCKBR RAT::P30553 | ||
| Residue: | 452 | ||
| Sequence: |
| ||
| BDBM82403 | |||
| n/a | |||
| Name | BDBM82403 | ||
| Synonyms: | CAS_108186 | CI-988 | NSC_108186 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C35H42N4O6 | ||
| Mol. Mass. | 614.7312 | ||
| SMILES | CC(Cc1c[nH]c2ccccc12)(NC(=O)OC1C2CC3CC(C2)CC1C3)C(=O)NCC(NC(=O)CCC(O)=O)c1ccccc1 |TLB:25:24:18.19.20:22,THB:15:16:18.19.20:22,20:19:16:21.22.23,20:21:16:18.19.25,25:19:22:16.23.24,(.76,2.29,;.32,.82,;-.11,-.66,;.95,-1.78,;.68,-3.29,;2.03,-4.02,;3.15,-2.96,;4.68,-3.07,;5.55,-1.8,;4.88,-.42,;3.35,-.3,;2.48,-1.57,;1.8,.38,;2.92,1.45,;2.55,2.94,;4.39,1.01,;5.51,2.08,;6.88,2.77,;6.88,4.17,;7.72,5.64,;9.09,4.95,;9.09,3.55,;8.25,2.08,;7.72,2.85,;6.35,3.55,;6.35,4.95,;-1.15,1.25,;-1.52,2.75,;-2.27,.19,;-3.75,.62,;-4.86,-.44,;-6.34,-.01,;-7.45,-1.07,;-7.09,-2.57,;-8.93,-.64,;-10.04,-1.7,;-11.52,-1.27,;-11.89,.23,;-12.64,-2.33,;-4.5,-1.94,;-3.02,-2.37,;-2.65,-3.87,;-3.77,-4.93,;-5.25,-4.5,;-5.61,-3,)| | ||
| Structure | 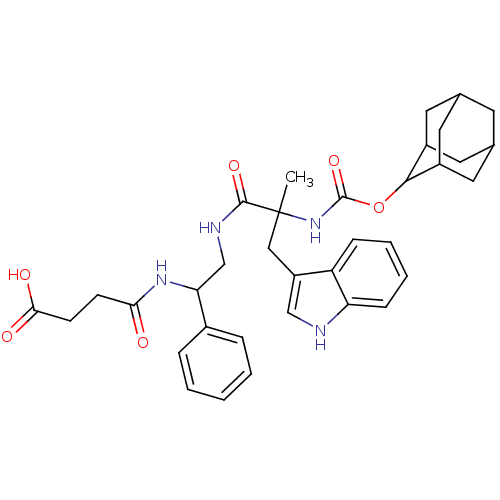
| ||