| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50011780 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1676701 (CHEMBL4026844) |
|---|
| Kd | 27600±n/a nM |
|---|
| Citation |  de Koning, MC; Joosen, MJA; Worek, F; Nachon, F; van Grol, M; Klaassen, SD; Alkema, DPW; Wille, T; de Bruijn, HM Application of the Ugi Multicomponent Reaction in the Synthesis of Reactivators of Nerve Agent Inhibited Acetylcholinesterase. J Med Chem60:9376-9392 (2017) [PubMed] Article de Koning, MC; Joosen, MJA; Worek, F; Nachon, F; van Grol, M; Klaassen, SD; Alkema, DPW; Wille, T; de Bruijn, HM Application of the Ugi Multicomponent Reaction in the Synthesis of Reactivators of Nerve Agent Inhibited Acetylcholinesterase. J Med Chem60:9376-9392 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_HUMAN | ACHE | Acetylcholinesterase (AChE) | Acetylcholinesterase (human AChE) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 67792.70 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P22303 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPQCLLHTPSLASPLLLLLLWLLGGGVGAEGREDAELLVTVRGGRLRGIRLKTPGGPV
SAFLGIPFAEPPMGPRRFLPPEPKQPWSGVVDATTFQSVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLVWIYGGGFYSGASSLDVYDGRFLVQAERTVLVSM
NYRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASV
GMHLLSPPSRGLFHRAVLQSGAPNGPWATVGMGEARRRATQLAHLVGCPPGGTGGNDTEL
VACLRTRPAQVLVNHEWHVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVG
VVKDEGSYFLVYGAPGFSKDNESLISRAEFLAGVRVGVPQVSDLAAEAVVLHYTDWLHPE
DPARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYVFEHRASTLSWPLWMGVPHGY
EIEFIFGIPLDPSRNYTAEEKIFAQRLMRYWANFARTGDPNEPRDPKAPQWPPYTAGAQQ
YVSLDLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQDRCSDL
|
|
|
|---|
| BDBM50011780 |
|---|
| n/a |
|---|
| Name | BDBM50011780 |
|---|
| Synonyms: | 2-[(E)-(hydroxyimino)methyl]-1-methylpyridinium chloride | CHEMBL748 | PRALIDOXIME | PRALIDOXIME CHLORIDE | Pralidoxine chloride |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C7H9N2O |
|---|
| Mol. Mass. | 137.1586 |
|---|
| SMILES | C[n+]1ccccc1CN=O |
|---|
| Structure | 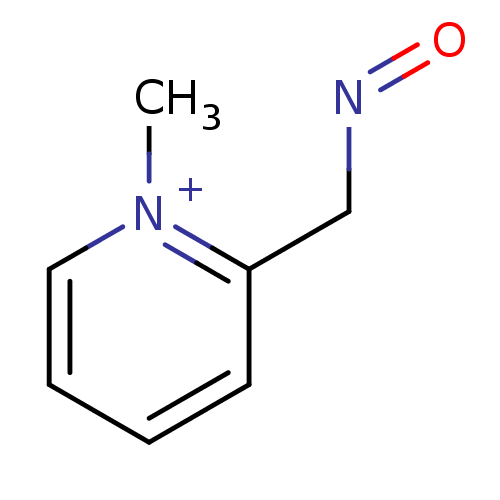
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 de Koning, MC; Joosen, MJA; Worek, F; Nachon, F; van Grol, M; Klaassen, SD; Alkema, DPW; Wille, T; de Bruijn, HM Application of the Ugi Multicomponent Reaction in the Synthesis of Reactivators of Nerve Agent Inhibited Acetylcholinesterase. J Med Chem60:9376-9392 (2017) [PubMed] Article
de Koning, MC; Joosen, MJA; Worek, F; Nachon, F; van Grol, M; Klaassen, SD; Alkema, DPW; Wille, T; de Bruijn, HM Application of the Ugi Multicomponent Reaction in the Synthesis of Reactivators of Nerve Agent Inhibited Acetylcholinesterase. J Med Chem60:9376-9392 (2017) [PubMed] Article