| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sucrase-isomaltase |
|---|
| Ligand | BDBM18355 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1672825 (CHEMBL4022854) |
|---|
| IC50 | 1500±n/a nM |
|---|
| Citation |  D'Alonzo, D; De Fenza, M; Porto, C; Iacono, R; Huebecker, M; Cobucci-Ponzano, B; Priestman, DA; Platt, F; Parenti, G; Moracci, M; Palumbo, G; Guaragna, A N-Butyl-l-deoxynojirimycin (l-NBDNJ): Synthesis of an Allosteric Enhancer of?-Glucosidase Activity for the Treatment of Pompe Disease. J Med Chem60:9462-9469 (2017) [PubMed] Article D'Alonzo, D; De Fenza, M; Porto, C; Iacono, R; Huebecker, M; Cobucci-Ponzano, B; Priestman, DA; Platt, F; Parenti, G; Moracci, M; Palumbo, G; Guaragna, A N-Butyl-l-deoxynojirimycin (l-NBDNJ): Synthesis of an Allosteric Enhancer of?-Glucosidase Activity for the Treatment of Pompe Disease. J Med Chem60:9462-9469 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sucrase-isomaltase |
|---|
| Name: | Sucrase-isomaltase |
|---|
| Synonyms: | n/a |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 209081.85 |
|---|
| Organism: | Mus musculus |
|---|
| Description: | ChEMBL_701783 |
|---|
| Residue: | 1818 |
|---|
| Sequence: | MAKKKFSGLEISLIVLFIIVTIIAIALVVVLATKVPAVEEVKSPTSTPSPGRCPPEQGEP
LNERINCIPEQHPTKAKCEERGCCWRPWNNTIIPWCFFADNHGYTAASVTNDNSGLKATL
SRIPSPTLFGEDIKSVLLTTQSQTRNRFRFKLTDPNNKRYEVPHQFVKDGNGIPAADTLY
DVKVSENPFSIKVIRKSNNKVLFDTSIGPLVYSNQYLQISTRLPSEYIYGFGEHIHKRFR
HDLYWKTWPIFTRDEIPGDNNHNLYGHQTFFMGIEDNSGKSYGVFLMNSNAMEVFIQPTP
IITYRVTGGVLDFYIFLGDTPEQVVQQYQELIGRPAMPAYWNLGFQLSRWNYVSLDKVKE
VVRRNREAGIPYDAQVTDIDYMEDKKDFTYDEVAFKGLPEFAQDLHNHGQKYIIILDPAI
SINKRANGAEYQTYVRGNEQNVWVKESDGTTSLIGEVWPGLTVYPDFTNPRTWEWWANEC
NLFHQQVEYDGLWIDMNEVSSFIHGSQKGCAPNLLNYPPFTPGILDKIMYSKTLCMDAVQ
HWGNQYDVHSLYGYSMAIATEKAVEKVFPNKRSFILTRSTFAGSGHHAAHWLGDNTASWE
QMEWSITGMLEFGMFGMPLVGADICGFLANTTEELCRRWMQLGAFYPFSRNHNAEGYAEQ
DPAFWGADSLLVNTSRHYLTIRYTLLPFLYTLFYRAHAFGETVARPFLHEFYEDPNSWIE
DTQFLWGPALLITPVLRPETKYVSAYIPDATWYDYETGEKRPWRKQRVDMYLPEDKIGLH
LRGGYIIPTQQPDVTTTASRKNPLGLIVALDENQAAKGELFWDDGESKDTIEKKIYILYE
FSVSNNNLIVNCTHSSYPEGNTLVFKTIKVLGLSATVTAVTVGENDQQMNPHLAFTFDAF
NKILSITDLTFNLGKTFIVRWTTQSFSDNEKFTCYPDVGTATEKTCVERGCIWEPVSGLA
NVPPCYFPSNHNPYLLTSTQKLATGITAELQLNPASARIKLPSNPISTLRVEVKYHKNDM
LQFKIYDAHHKRYEVPVPLNIPDTPTSSEENRLYDVEIKENPFGIQVRRRSTGKLIWDSC
LPGFAFNDQFIQISTRLPSQYLYGFGEAEHTAFKRNLNWHTWGMFTRDQPPGYKLNSYGF
HPYYMALEDEGNAHGVLLQNSNGMDVTFQPTPALTYRTIGGILDFYMFLGPTPEGATKQY
HEVIGFPVMPPYWALGFQLCRYGYRNTSEIEQLYNDMKAAQIPYDVQYTDINYMERQLDF
TIGERFKTLPQFVEKIRKEGMKYIVILDPAISGNETQPYPAFERGIQKDVFVKWPNTNDI
CWAKVWPDLPNITIDETITEDEAVNASRAHVAFPDFFRNSTSEWWTREIYDFYNEKMKFD
GLWIDMNEPSSFVNGTVTNKCRNDTLNYPPYFPELTKRNEGLHFRTMCMETEHILSDGSS
VLHYDVHNLYGWSQVKPTLDALRNTTGLRGIVISRSTYPTAGRWGGHWLGDNYANWENLE
KSLIGMLEFNLFGIPYVGADICGFFNDSEYHLCARWMQVGAFYPYSRNHNIQFTRRQDPV
SWNETFAQMSKKVLEIRYTLLPYFYTQMHEAHIHGGTVIRPLMHEFFDDKETWEIYKQFL
WGPAFMVTPVIGPFQTAVNGYVPKARWFDYHTGEDIKVRGKLQTFSAPFDTINLHVRGGY
ILPCQEPAQNTYYSRQNYMKLIVAADDNQTAKGSLFWDDGESIDTYEKNQYTLIEFNLNQ
KTLTSTVLKNGYRNKSEMKLGNIYVWGKGTTHINQVNLTYGGNEQQLLFTQDEAKEILTI
ELKNVNVTLDEPIQISWS
|
|
|
|---|
| BDBM18355 |
|---|
| n/a |
|---|
| Name | BDBM18355 |
|---|
| Synonyms: | (2R,3R,4R,5S)-1-butyl-2-(hydroxymethyl)piperidine-3,4,5-triol | CHEMBL1029 | MIGLUSTAT | N-Butyl-DNJ | US20230339856, Compound NB-DNJ | US9181184, 5 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C10H21NO4 |
|---|
| Mol. Mass. | 219.278 |
|---|
| SMILES | CCCCN1C[C@H](O)[C@@H](O)[C@H](O)[C@H]1CO |
|---|
| Structure | 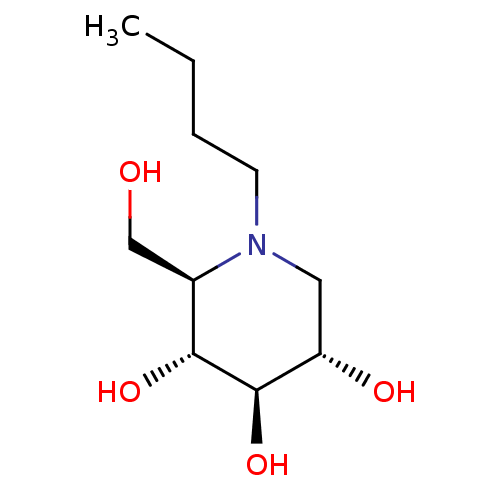
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 D'Alonzo, D; De Fenza, M; Porto, C; Iacono, R; Huebecker, M; Cobucci-Ponzano, B; Priestman, DA; Platt, F; Parenti, G; Moracci, M; Palumbo, G; Guaragna, A N-Butyl-l-deoxynojirimycin (l-NBDNJ): Synthesis of an Allosteric Enhancer of?-Glucosidase Activity for the Treatment of Pompe Disease. J Med Chem60:9462-9469 (2017) [PubMed] Article
D'Alonzo, D; De Fenza, M; Porto, C; Iacono, R; Huebecker, M; Cobucci-Ponzano, B; Priestman, DA; Platt, F; Parenti, G; Moracci, M; Palumbo, G; Guaragna, A N-Butyl-l-deoxynojirimycin (l-NBDNJ): Synthesis of an Allosteric Enhancer of?-Glucosidase Activity for the Treatment of Pompe Disease. J Med Chem60:9462-9469 (2017) [PubMed] Article