| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1B1 |
|---|
| Ligand | BDBM50344942 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1684888 (CHEMBL4035367) |
|---|
| IC50 | 40000±n/a nM |
|---|
| Citation |  Dutour, R; Poirier, D Inhibitors of cytochrome P450 (CYP) 1B1. Eur J Med Chem135:296-306 (2017) [PubMed] Article Dutour, R; Poirier, D Inhibitors of cytochrome P450 (CYP) 1B1. Eur J Med Chem135:296-306 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1B1 |
|---|
| Name: | Cytochrome P450 1B1 |
|---|
| Synonyms: | CP1B1_HUMAN | CYP1B1 | CYPIB1 | Cytochrome P450 1B1 (CYP1B1) |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 60861.81 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1474523 |
|---|
| Residue: | 543 |
|---|
| Sequence: | MGTSLSPNDPWPLNPLSIQQTTLLLLLSVLATVHVGQRLLRQRRRQLRSAPPGPFAWPLI
GNAAAVGQAAHLSFARLARRYGDVFQIRLGSCPIVVLNGERAIHQALVQQGSAFADRPAF
ASFRVVSGGRSMAFGHYSEHWKVQRRAAHSMMRNFFTRQPRSRQVLEGHVLSEARELVAL
LVRGSADGAFLDPRPLTVVAVANVMSAVCFGCRYSHDDPEFRELLSHNEEFGRTVGAGSL
VDVMPWLQYFPNPVRTVFREFEQLNRNFSNFILDKFLRHCESLRPGAAPRDMMDAFILSA
EKKAAGDSHGGGARLDLENVPATITDIFGASQDTLSTALQWLLLLFTRYPDVQTRVQAEL
DQVVGRDRLPCMGDQPNLPYVLAFLYEAMRFSSFVPVTIPHATTANTSVLGYHIPKDTVV
FVNQWSVNHDPLKWPNPENFDPARFLDKDGLINKDLTSRVMIFSVGKRRCIGEELSKMQL
FLFISILAHQCDFRANPNEPAKMNFSYGLTIKPKSFKVNVTLRESMELLDSAVQNLQAKE
TCQ
|
|
|
|---|
| BDBM50344942 |
|---|
| n/a |
|---|
| Name | BDBM50344942 |
|---|
| Synonyms: | CHEMBL532 | E-MYCIN E | ERYTHROMYCIN | ERYTHROMYCIN ETHYLSUCCINATE | Erycette | Erymax | Erythromycin A | Erythromycin C | Ilotycin | T-Stat |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C37H67NO13 |
|---|
| Mol. Mass. | 733.9268 |
|---|
| SMILES | CC[C@H]1OC(=O)[C@H](C)[C@@H](O[C@H]2C[C@@](C)(OC)[C@@H](O)[C@H](C)O2)[C@H](C)[C@@H](O[C@@H]2O[C@H](C)C[C@@H]([C@H]2O)N(C)C)[C@](C)(O)C[C@@H](C)C(=O)[C@H](C)[C@@H](O)[C@]1(C)O |r| |
|---|
| Structure | 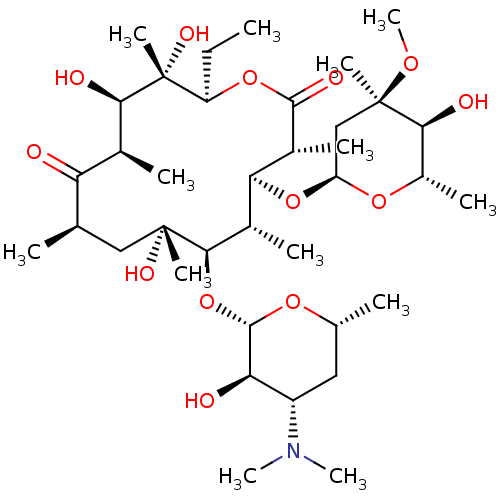
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dutour, R; Poirier, D Inhibitors of cytochrome P450 (CYP) 1B1. Eur J Med Chem135:296-306 (2017) [PubMed] Article
Dutour, R; Poirier, D Inhibitors of cytochrome P450 (CYP) 1B1. Eur J Med Chem135:296-306 (2017) [PubMed] Article