| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Proto-oncogene tyrosine-protein kinase Src |
|---|
| Ligand | BDBM50252727 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1691362 (CHEMBL4042011) |
|---|
| IC50 | <0.500000±n/a nM |
|---|
| Citation |  Gaboriaud-Kolar, N; Myrianthopoulos, V; Vougogiannopoulou, K; Gerolymatos, P; Horne, DA; Jove, R; Mikros, E; Nam, S; Skaltsounis, AL Natural-Based Indirubins Display Potent Cytotoxicity toward Wild-Type and T315I-Resistant Leukemia Cell Lines. J Nat Prod79:2464-2471 (2016) [PubMed] Article Gaboriaud-Kolar, N; Myrianthopoulos, V; Vougogiannopoulou, K; Gerolymatos, P; Horne, DA; Jove, R; Mikros, E; Nam, S; Skaltsounis, AL Natural-Based Indirubins Display Potent Cytotoxicity toward Wild-Type and T315I-Resistant Leukemia Cell Lines. J Nat Prod79:2464-2471 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Proto-oncogene tyrosine-protein kinase Src |
|---|
| Name: | Proto-oncogene tyrosine-protein kinase Src |
|---|
| Synonyms: | Calmodulin/Proto-oncogene tyrosine-protein kinase Src | Protein cereblon/Tyrosine-protein kinase SRC | Proto-oncogene c-Src | Proto-oncogene tyrosine-protein kinase Src (c-Src) | SRC | SRC1 | SRC_HUMAN | Tyrosine-protein kinase Src (SRC) | V-src sarcoma (Schmidt-Ruppin A-2) viral oncogene homolog (avian) | c-Src | p60-Src | pp60c-src |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 59838.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P12931 |
|---|
| Residue: | 536 |
|---|
| Sequence: | MGSNKSKPKDASQRRRSLEPAENVHGAGGGAFPASQTPSKPASADGHRGPSAAFAPAAAE
PKLFGGFNSSDTVTSPQRAGPLAGGVTTFVALYDYESRTETDLSFKKGERLQIVNNTEGD
WWLAHSLSTGQTGYIPSNYVAPSDSIQAEEWYFGKITRRESERLLLNAENPRGTFLVRES
ETTKGAYCLSVSDFDNAKGLNVKHYKIRKLDSGGFYITSRTQFNSLQQLVAYYSKHADGL
CHRLTTVCPTSKPQTQGLAKDAWEIPRESLRLEVKLGQGCFGEVWMGTWNGTTRVAIKTL
KPGTMSPEAFLQEAQVMKKLRHEKLVQLYAVVSEEPIYIVTEYMSKGSLLDFLKGETGKY
LRLPQLVDMAAQIASGMAYVERMNYVHRDLRAANILVGENLVCKVADFGLARLIEDNEYT
ARQGAKFPIKWTAPEAALYGRFTIKSDVWSFGILLTELTTKGRVPYPGMVNREVLDQVER
GYRMPCPPECPESLHDLMCQCWRKEPEERPTFEYLQAFLEDYFTSTEPQYQPGENL
|
|
|
|---|
| BDBM50252727 |
|---|
| n/a |
|---|
| Name | BDBM50252727 |
|---|
| Synonyms: | (2'Z-3'E)-6-Bromoindirubin-3'-(O-{2-[N-methyl,N-(2,3-dihydroxypropyl)amino]ethyl}oxime] | CHEMBL494867 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H23BrN4O4 |
|---|
| Mol. Mass. | 487.346 |
|---|
| SMILES | CN(CCO\N=C1\C(\Nc2ccccc\12)=C1\C(=O)Nc2cc(Br)ccc12)CC(O)CO |
|---|
| Structure | 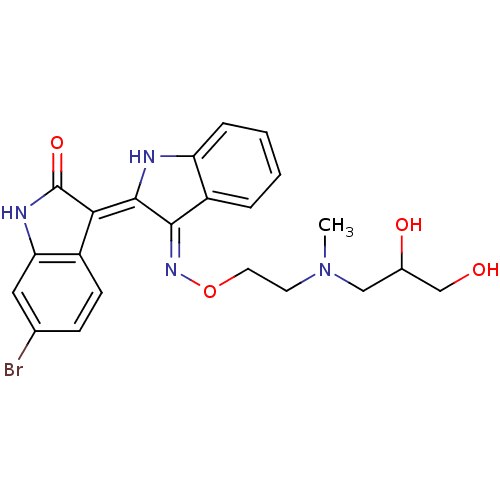
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gaboriaud-Kolar, N; Myrianthopoulos, V; Vougogiannopoulou, K; Gerolymatos, P; Horne, DA; Jove, R; Mikros, E; Nam, S; Skaltsounis, AL Natural-Based Indirubins Display Potent Cytotoxicity toward Wild-Type and T315I-Resistant Leukemia Cell Lines. J Nat Prod79:2464-2471 (2016) [PubMed] Article
Gaboriaud-Kolar, N; Myrianthopoulos, V; Vougogiannopoulou, K; Gerolymatos, P; Horne, DA; Jove, R; Mikros, E; Nam, S; Skaltsounis, AL Natural-Based Indirubins Display Potent Cytotoxicity toward Wild-Type and T315I-Resistant Leukemia Cell Lines. J Nat Prod79:2464-2471 (2016) [PubMed] Article