| Reaction Details |
|---|
 | Report a problem with these data |
| Target | DNA gyrase subunit B |
|---|
| Ligand | BDBM50408191 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1739367 (CHEMBL4155117) |
|---|
| IC50 | 13±n/a nM |
|---|
| Citation |  Durcik, M; Lovison, D; Skok, ?; Durante Cruz, C; Tammela, P; Toma?i?, T; Benedetto Tiz, D; Draskovits, G; Nyerges, �; P�l, C; Ila?, J; Peterlin Ma?i?, L; Kikelj, D; Zidar, N New N-phenylpyrrolamide DNA gyrase B inhibitors: Optimization of efficacy and antibacterial activity. Eur J Med Chem154:117-132 (2018) [PubMed] Article Durcik, M; Lovison, D; Skok, ?; Durante Cruz, C; Tammela, P; Toma?i?, T; Benedetto Tiz, D; Draskovits, G; Nyerges, �; P�l, C; Ila?, J; Peterlin Ma?i?, L; Kikelj, D; Zidar, N New N-phenylpyrrolamide DNA gyrase B inhibitors: Optimization of efficacy and antibacterial activity. Eur J Med Chem154:117-132 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| DNA gyrase subunit B |
|---|
| Name: | DNA gyrase subunit B |
|---|
| Synonyms: | DNA gyrase subunit B | DNA gyrase subunit B (gyrB) | GYRB_ECOLI | Type IIA topoisomerase subunit GyrB | acrB | cou | gyrB | himB | hisU | nalC | parA | pcbA |
|---|
| Type: | Enzyme Subunit |
|---|
| Mol. Mass.: | 89941.28 |
|---|
| Organism: | Escherichia coli (strain K12) |
|---|
| Description: | P0AES6 |
|---|
| Residue: | 804 |
|---|
| Sequence: | MSNSYDSSSIKVLKGLDAVRKRPGMYIGDTDDGTGLHHMVFEVVDNAIDEALAGHCKEII
VTIHADNSVSVQDDGRGIPTGIHPEEGVSAAEVIMTVLHAGGKFDDNSYKVSGGLHGVGV
SVVNALSQKLELVIQREGKIHRQIYEHGVPQAPLAVTGETEKTGTMVRFWPSLETFTNVT
EFEYEILAKRLRELSFLNSGVSIRLRDKRDGKEDHFHYEGGIKAFVEYLNKNKTPIHPNI
FYFSTEKDGIGVEVALQWNDGFQENIYCFTNNIPQRDGGTHLAGFRAAMTRTLNAYMDKE
GYSKKAKVSATGDDAREGLIAVVSVKVPDPKFSSQTKDKLVSSEVKSAVEQQMNELLAEY
LLENPTDAKIVVGKIIDAARAREAARRAREMTRRKGALDLAGLPGKLADCQERDPALSEL
YLVEGDSAGGSAKQGRNRKNQAILPLKGKILNVEKARFDKMLSSQEVATLITALGCGIGR
DEYNPDKLRYHSIIIMTDADVDGSHIRTLLLTFFYRQMPEIVERGHVYIAQPPLYKVKKG
KQEQYIKDDEAMDQYQISIALDGATLHTNASAPALAGEALEKLVSEYNATQKMINRMERR
YPKAMLKELIYQPTLTEADLSDEQTVTRWVNALVSELNDKEQHGSQWKFDVHTNAEQNLF
EPIVRVRTHGVDTDYPLDHEFITGGEYRRICTLGEKLRGLLEEDAFIERGERRQPVASFE
QALDWLVKESRRGLSIQRYKGLGEMNPEQLWETTMDPESRRMLRVTVKDAIAADQLFTTL
MGDAVEPRRAFIEENALKAANIDI
|
|
|
|---|
| BDBM50408191 |
|---|
| n/a |
|---|
| Name | BDBM50408191 |
|---|
| Synonyms: | CHEMBL4163243 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H20Cl2N6O5 |
|---|
| Mol. Mass. | 483.305 |
|---|
| SMILES | C[C@H](NC(=O)c1ccc(NC(=O)c2[nH]c(C)c(Cl)c2Cl)c(OCCN)c1)c1n[nH]c(=O)o1 |r| |
|---|
| Structure | 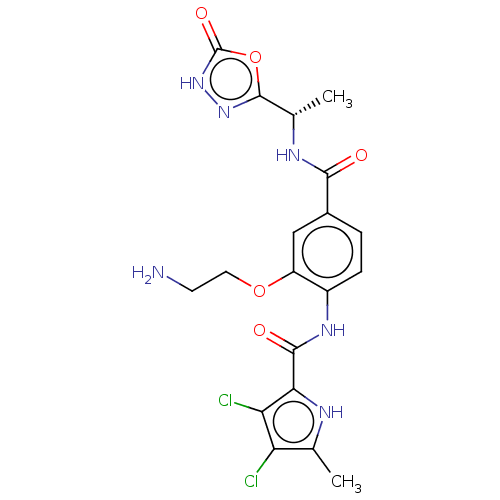
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Durcik, M; Lovison, D; Skok, ?; Durante Cruz, C; Tammela, P; Toma?i?, T; Benedetto Tiz, D; Draskovits, G; Nyerges, �; P�l, C; Ila?, J; Peterlin Ma?i?, L; Kikelj, D; Zidar, N New N-phenylpyrrolamide DNA gyrase B inhibitors: Optimization of efficacy and antibacterial activity. Eur J Med Chem154:117-132 (2018) [PubMed] Article
Durcik, M; Lovison, D; Skok, ?; Durante Cruz, C; Tammela, P; Toma?i?, T; Benedetto Tiz, D; Draskovits, G; Nyerges, �; P�l, C; Ila?, J; Peterlin Ma?i?, L; Kikelj, D; Zidar, N New N-phenylpyrrolamide DNA gyrase B inhibitors: Optimization of efficacy and antibacterial activity. Eur J Med Chem154:117-132 (2018) [PubMed] Article