| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bifunctional dihydrofolate reductase-thymidylate synthase |
|---|
| Ligand | BDBM50035483 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1747167 (CHEMBL4181677) |
|---|
| IC50 | 0.390000±n/a nM |
|---|
| Citation |  Chen, W; Huang, Z; Wang, W; Mao, F; Guan, L; Tang, Y; Jiang, H; Li, J; Huang, J; Jiang, L; Zhu, J Discovery of new antimalarial agents: Second-generation dual inhibitors against FP-2 and PfDHFR via fragments assembely. Bioorg Med Chem25:6467-6478 (2017) [PubMed] Article Chen, W; Huang, Z; Wang, W; Mao, F; Guan, L; Tang, Y; Jiang, H; Li, J; Huang, J; Jiang, L; Zhu, J Discovery of new antimalarial agents: Second-generation dual inhibitors against FP-2 and PfDHFR via fragments assembely. Bioorg Med Chem25:6467-6478 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bifunctional dihydrofolate reductase-thymidylate synthase |
|---|
| Name: | Bifunctional dihydrofolate reductase-thymidylate synthase |
|---|
| Synonyms: | DHFR-TS | DRTS_PLAFK | Dihydrofolate Reductase-Thymidylate Synthase (DHFR-TS) Mutant KICB1 | Dihydrofolate reductase | PfDHFR-TS double mutant (C59R+S108N) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 71822.51 |
|---|
| Organism: | Plasmodium falciparum (isolate K1 / Thailand) |
|---|
| Description: | The mutant clone was prepared by cassette mutagenesis using wildtype pfDHFR as a template, and expressed in E. coli. |
|---|
| Residue: | 608 |
|---|
| Sequence: | MMEQVCDVFDIYAICACCKVESKNEGKKNEVFNNYTFRGLGNKGVLPWKCNSLDMKYFRA
VTTYVNESKYEKLKYKRCKYLNKETVDNVNDMPNSKKLQNVVVMGRTNWESIPKKFKPLS
NRINVILSRTLKKEDFDEDVYIINKVEDLIVLLGKLNYYKCFIIGGSVVYQEFLEKKLIK
KIYFTRINSTYECDVFFPEINENEYQIISVSDVYTSNNTTLDFIIYKKTNNKMLNEQNCI
KGEEKNNDMPLKNDDKDTCHMKKLTEFYKNVDKYKINYENDDDDEEEDDFVYFNFNKEKE
EKNKNSIHPNDFQIYNSLKYKYHPEYQYLNIIYDIMMNGNKQSDRTGVGVLSKFGYIMKF
DLSQYFPLLTTKKLFLRGIIEELLWFIRGETNGNTLLNKNVRIWEANGTREFLDNRKLFH
REVNDLGPIYGFQWRHFGAEYTNMYDNYENKGVDQLKNIINLIKNDPTSRRILLCAWNVK
DLDQMALPPCHILCQFYVFDGKLSCIMYQRSCDLGLGVPFNIASYSIFTHMIAQVCNLQP
AQFIHVLGNAHVYNNHIDSLKIQLNRIPYPFPTLKLNPDIKNIEDFTISDFTIQNYVHHE
KISMDMAA
|
|
|
|---|
| BDBM50035483 |
|---|
| n/a |
|---|
| Name | BDBM50035483 |
|---|
| Synonyms: | 5-Chloro-N*6*-(2,5-dimethoxy-benzyl)-quinazoline-2,4,6-triamine | 5-chloro-N6-(2,5-dimethoxybenzyl)quinazoline-2,4,6-triamine | 5-chloro-N~6~-(2,5-dimethoxybenzyl)quinazoline-2,4,6-triamine | CHEMBL138060 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H18ClN5O2 |
|---|
| Mol. Mass. | 359.81 |
|---|
| SMILES | COc1ccc(OC)c(CNc2ccc3nc(N)nc(N)c3c2Cl)c1 |
|---|
| Structure | 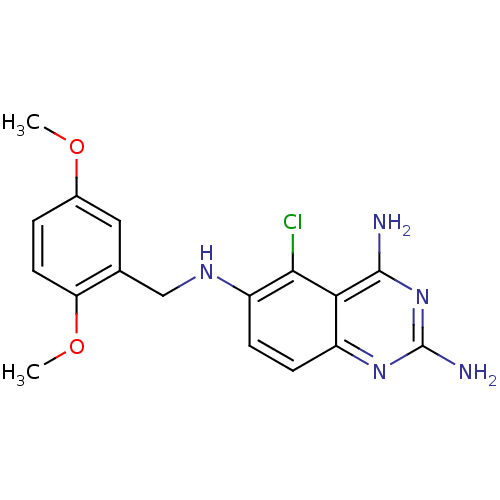
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chen, W; Huang, Z; Wang, W; Mao, F; Guan, L; Tang, Y; Jiang, H; Li, J; Huang, J; Jiang, L; Zhu, J Discovery of new antimalarial agents: Second-generation dual inhibitors against FP-2 and PfDHFR via fragments assembely. Bioorg Med Chem25:6467-6478 (2017) [PubMed] Article
Chen, W; Huang, Z; Wang, W; Mao, F; Guan, L; Tang, Y; Jiang, H; Li, J; Huang, J; Jiang, L; Zhu, J Discovery of new antimalarial agents: Second-generation dual inhibitors against FP-2 and PfDHFR via fragments assembely. Bioorg Med Chem25:6467-6478 (2017) [PubMed] Article