| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 3-hydroxy-3-methylglutaryl-coenzyme A reductase |
|---|
| Ligand | BDBM50020290 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_365 |
|---|
| IC50 | 1.6±n/a nM |
|---|
| Citation |  Heathcock, CH; Davis, BR; Hadley, CR Synthesis and biological evaluation of a monocyclic, fully functional analogue of compactin. J Med Chem32:197-202 (1989) [PubMed] Heathcock, CH; Davis, BR; Hadley, CR Synthesis and biological evaluation of a monocyclic, fully functional analogue of compactin. J Med Chem32:197-202 (1989) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase |
|---|
| Name: | 3-hydroxy-3-methylglutaryl-coenzyme A reductase |
|---|
| Synonyms: | HMDH_RAT | HMG-CoA reductase | Hmgcr |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 96689.85 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Isolated rat liver microsomes were used as enzyme source. |
|---|
| Residue: | 887 |
|---|
| Sequence: | MLSRLFRMHGLFVASHPWEVIVGTVTLTICMMSMNMFTGNNKICGWNYECPKFEEDVLSS
DIIILTITRCIAILYIYFQFQNLRQLGSKYILGIAGLFTIFSSFVFSTVVIHFLDKELTG
LNEALPFFLLLIDLSRASALAKFALSSNSQDEVRENIARGMAILGPTFTLDALVECLVIG
VGTMSGVRQLEIMCCFGCMSVLANYFVFMTFFPACVSLVLELSRESREGRPIWQLSHFAR
VLEEEENKPNPVTQRVKMIMSLGLVLVHAHSRWIADPSPQNSTAEQSKVSLGLAEDVSKR
IEPSVSLWQFYLSKMISMDIEQVITLSLALLLAVKYIFFEQAETESTLSLKNPITSPVVT
PKKAQDNCCRREPLLVRRNQKLSSVEEDPGVNQDRKVEVIKPLVAEAETSGRATFVLGAS
AASPPLALGAQEPGIELPSEPRPNEECLQILESAEKGAKFLSDAEIIQLVNAKHIPAYKL
ETLMETHERGVSIRRQLLSAKLAEPSSLQYLPYRDYNYSLVMGACCENVIGYMPIPVGVA
GPLCLDGKEYQVPMATTEGCLVASTNRGCRAISLGGGASSRVLADGMSRGPVVRLPRACD
SAEVKSWLETPEGFAVVKEAFDSTSRFARLQKLHVTLAGRNLYIRLQSKTGDAMGMNMIS
KGTEKALLKLQEFFPELQILAVSGNYCTDKKPAAINWIEGRGKTVVCEAVIPAKVVREVL
KTTTEAMVDVNINKNLVGSAMAGSIGGYNAHAANIVTAIYIACGQDAAQNVGSSNCITLM
EASGPTNEDLYISCTMPSIEIGTVGGGTNLLPQQACLQMLGVQGACKDNPGENARQLARI
VCGTVMAGELSLMAALAAGHLVRSHMVHNRSKINLQDLQGTCTKKAA
|
|
|
|---|
| BDBM50020290 |
|---|
| n/a |
|---|
| Name | BDBM50020290 |
|---|
| Synonyms: | 7-[2,6-Dimethyl-8-(2-methyl-butyryloxy)-1,2,4a,5,6,7,8,8a-octahydro-naphthalen-1-yl]-3,5-dihydroxy-heptanoic acid | CHEMBL59166 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H40O6 |
|---|
| Mol. Mass. | 424.5708 |
|---|
| SMILES | CC[C@H](C)C(=O)O[C@H]1C[C@@H](C)C[C@@H]2C=C[C@H](C)[C@H](CCC(O)C[C@@H](O)CC(O)=O)C12 |c:13| |
|---|
| Structure | 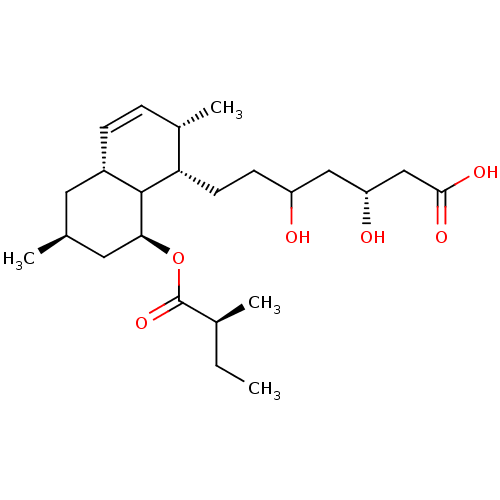
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Heathcock, CH; Davis, BR; Hadley, CR Synthesis and biological evaluation of a monocyclic, fully functional analogue of compactin. J Med Chem32:197-202 (1989) [PubMed]
Heathcock, CH; Davis, BR; Hadley, CR Synthesis and biological evaluation of a monocyclic, fully functional analogue of compactin. J Med Chem32:197-202 (1989) [PubMed]