| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Platelet-activating factor acetylhydrolase |
|---|
| Ligand | BDBM50125265 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1752778 (CHEMBL4187538) |
|---|
| IC50 | 0.250000±n/a nM |
|---|
| Citation |  Guibbal, F; B�nard, S; Patch�, J; Meneyrol, V; Couprie, J; Yong-Sang, J; Meilhac, O; Jestin, E Regioselectivity of thiouracil alkylation: Application to optimization of Darapladib synthesis. Bioorg Med Chem Lett28:787-792 (2018) [PubMed] Article Guibbal, F; B�nard, S; Patch�, J; Meneyrol, V; Couprie, J; Yong-Sang, J; Meilhac, O; Jestin, E Regioselectivity of thiouracil alkylation: Application to optimization of Darapladib synthesis. Bioorg Med Chem Lett28:787-792 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Platelet-activating factor acetylhydrolase |
|---|
| Name: | Platelet-activating factor acetylhydrolase |
|---|
| Synonyms: | 1-alkyl-2-acetylglycerophosphocholine esterase | 2-acetyl-1-alkylglycerophosphocholine esterase | LDL-PLA(2) | LDL-associated phospholipase A2 | PAF 2-acylhydrolase | PAF acetylhydrolase | PAFAH | PAFA_HUMAN | PLA2G7 | Platelet-activating factor acetylhydrolase |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 50084.41 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_156212 |
|---|
| Residue: | 441 |
|---|
| Sequence: | MVPPKLHVLFCLCGCLAVVYPFDWQYINPVAHMKSSAWVNKIQVLMAAASFGQTKIPRGN
GPYSVGCTDLMFDHTNKGTFLRLYYPSQDNDRLDTLWIPNKEYFWGLSKFLGTHWLMGNI
LRLLFGSMTTPANWNSPLRPGEKYPLVVFSHGLGAFRTLYSAIGIDLASHGFIVAAVEHR
DRSASATYYFKDQSAAEIGDKSWLYLRTLKQEEETHIRNEQVRQRAKECSQALSLILDID
HGKPVKNALDLKFDMEQLKDSIDREKIAVIGHSFGGATVIQTLSEDQRFRCGIALDAWMF
PLGDEVYSRIPQPLFFINSEYFQYPANIIKMKKCYSPDKERKMITIRGSVHQNFADFTFA
TGKIIGHMLKLKGDIDSNVAIDLSNKASLAFLQKHLGLHKDFDQWDCLIEGDDENLIPGT
NINTTNQHIMLQNSSGIEKYN
|
|
|
|---|
| BDBM50125265 |
|---|
| n/a |
|---|
| Name | BDBM50125265 |
|---|
| Synonyms: | CHEMBL204021 | N-(2-Diethylamino-ethyl)-2-[2-(4-fluoro-benzylsulfanyl)-4-oxo-4,5,6,7-tetrahydro-cyclopentapyrimidin-1-yl]-N-(4'-trifluoromethyl-biphenyl-4-ylmethyl)-acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C36H38F4N4O2S |
|---|
| Mol. Mass. | 666.771 |
|---|
| SMILES | CCN(CC)CCN(Cc1ccc(cc1)-c1ccc(cc1)C(F)(F)F)C(=O)Cn1c2CCCc2c(=O)nc1SCc1ccc(F)cc1 |
|---|
| Structure | 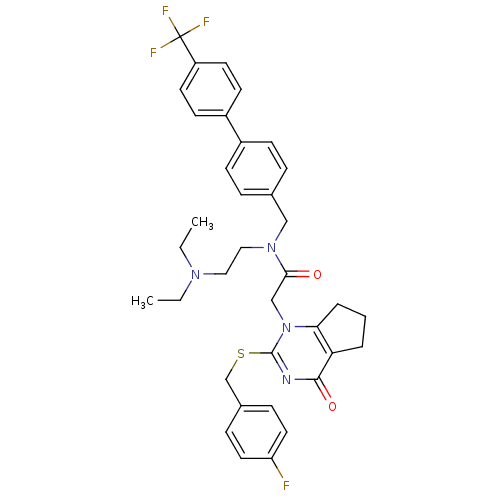
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Guibbal, F; B�nard, S; Patch�, J; Meneyrol, V; Couprie, J; Yong-Sang, J; Meilhac, O; Jestin, E Regioselectivity of thiouracil alkylation: Application to optimization of Darapladib synthesis. Bioorg Med Chem Lett28:787-792 (2018) [PubMed] Article
Guibbal, F; B�nard, S; Patch�, J; Meneyrol, V; Couprie, J; Yong-Sang, J; Meilhac, O; Jestin, E Regioselectivity of thiouracil alkylation: Application to optimization of Darapladib synthesis. Bioorg Med Chem Lett28:787-792 (2018) [PubMed] Article