| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glucocorticoid receptor |
|---|
| Ligand | BDBM50354850 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1755053 (CHEMBL4189813) |
|---|
| Ki | 1.3±n/a nM |
|---|
| Citation |  Ghidini, E; Marchini, G; Capelli, AM; Carnini, C; Cenacchi, V; Fioni, A; Facchinetti, F; Rancati, F Novel Pyrrolidine Derivatives of Budesonide as Long Acting Inhaled Corticosteroids for the Treatment of Pulmonary Inflammatory Diseases. J Med Chem61:4757-4773 (2018) [PubMed] Article Ghidini, E; Marchini, G; Capelli, AM; Carnini, C; Cenacchi, V; Fioni, A; Facchinetti, F; Rancati, F Novel Pyrrolidine Derivatives of Budesonide as Long Acting Inhaled Corticosteroids for the Treatment of Pulmonary Inflammatory Diseases. J Med Chem61:4757-4773 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glucocorticoid receptor |
|---|
| Name: | Glucocorticoid receptor |
|---|
| Synonyms: | GCR_HUMAN | GR | GRL | Glucocorticoid | Glucocorticoid receptor (GRFP) | NR3C1 | Nuclear receptor subfamily 3 group C member 1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 85656.87 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P04150 |
|---|
| Residue: | 777 |
|---|
| Sequence: | MDSKESLTPGREENPSSVLAQERGDVMDFYKTLRGGATVKVSASSPSLAVASQSDSKQRR
LLVDFPKGSVSNAQQPDLSKAVSLSMGLYMGETETKVMGNDLGFPQQGQISLSSGETDLK
LLEESIANLNRSTSVPENPKSSASTAVSAAPTEKEFPKTHSDVSSEQQHLKGQTGTNGGN
VKLYTTDQSTFDILQDLEFSSGSPGKETNESPWRSDLLIDENCLLSPLAGEDDSFLLEGN
SNEDCKPLILPDTKPKIKDNGDLVLSSPSNVTLPQVKTEKEDFIELCTPGVIKQEKLGTV
YCQASFPGANIIGNKMSAISVHGVSTSGGQMYHYDMNTASLSQQQDQKPIFNVIPPIPVG
SENWNRCQGSGDDNLTSLGTLNFPGRTVFSNGYSSPSMRPDVSSPPSSSSTATTGPPPKL
CLVCSDEASGCHYGVLTCGSCKVFFKRAVEGQHNYLCAGRNDCIIDKIRRKNCPACRYRK
CLQAGMNLEARKTKKKIKGIQQATTGVSQETSENPGNKTIVPATLPQLTPTLVSLLEVIE
PEVLYAGYDSSVPDSTWRIMTTLNMLGGRQVIAAVKWAKAIPGFRNLHLDDQMTLLQYSW
MFLMAFALGWRSYRQSSANLLCFAPDLIINEQRMTLPCMYDQCKHMLYVSSELHRLQVSY
EEYLCMKTLLLLSSVPKDGLKSQELFDEIRMTYIKELGKAIVKREGNSSQNWQRFYQLTK
LLDSMHEVVENLLNYCFQTFLDKTMSIEFPEMLAEIITNQIPKYSNGNIKKLLFHQK
|
|
|
|---|
| BDBM50354850 |
|---|
| n/a |
|---|
| Name | BDBM50354850 |
|---|
| Synonyms: | BUDESONIDE | US10869929, Compound Budesonide | US11554172, Compound Budesonide | US11760775, Compound Budesonide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H34O6 |
|---|
| Mol. Mass. | 430.5339 |
|---|
| SMILES | CCCC1O[C@@H]2C[C@H]3[C@@H]4CCC5=CC(=O)C=C[C@]5(C)[C@H]4[C@@H](O)C[C@]3(C)[C@@]2(O1)C(=O)CO |r,c:15,t:11| |
|---|
| Structure | 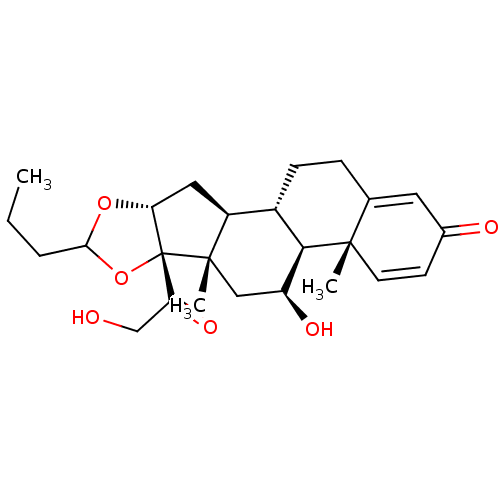
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ghidini, E; Marchini, G; Capelli, AM; Carnini, C; Cenacchi, V; Fioni, A; Facchinetti, F; Rancati, F Novel Pyrrolidine Derivatives of Budesonide as Long Acting Inhaled Corticosteroids for the Treatment of Pulmonary Inflammatory Diseases. J Med Chem61:4757-4773 (2018) [PubMed] Article
Ghidini, E; Marchini, G; Capelli, AM; Carnini, C; Cenacchi, V; Fioni, A; Facchinetti, F; Rancati, F Novel Pyrrolidine Derivatives of Budesonide as Long Acting Inhaled Corticosteroids for the Treatment of Pulmonary Inflammatory Diseases. J Med Chem61:4757-4773 (2018) [PubMed] Article