| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Carnitine O-palmitoyltransferase 1, liver isoform |
|---|
| Ligand | BDBM50024430 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_216362 |
|---|
| IC50 | 20±n/a nM |
|---|
| Citation |  Ho, W; Tarhan, O; Kiorpes, TC; Tutwiler, GF; Mohrbacher, RJ Resolution of (+/-)-2-tetradecyloxiranecarboxylic acid. Absolute configuration and chiral synthesis of the hypoglycemic R enantiomer and biological activity of enantiomers. J Med Chem30:1094-7 (1987) [PubMed] Ho, W; Tarhan, O; Kiorpes, TC; Tutwiler, GF; Mohrbacher, RJ Resolution of (+/-)-2-tetradecyloxiranecarboxylic acid. Absolute configuration and chiral synthesis of the hypoglycemic R enantiomer and biological activity of enantiomers. J Med Chem30:1094-7 (1987) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Carnitine O-palmitoyltransferase 1, liver isoform |
|---|
| Name: | Carnitine O-palmitoyltransferase 1, liver isoform |
|---|
| Synonyms: | CPT I | CPT1-L | CPT1A_RAT | CPTI-L | Carnitine O-palmitoyltransferase I, liver isoform | Carnitine palmitoyltransferase 1A | Cpt-1 | Cpt1 | Cpt1a |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 88147.11 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_887445 |
|---|
| Residue: | 773 |
|---|
| Sequence: | MAEAHQAVAFQFTVTPDGIDLRLSHEALKQICLSGLHSWKKKFIRFKNGIITGVFPANPS
SWLIVVVGVISSMHAKVDPSLGMIAKISRTLDTTGRMSSQTKNIVSGVLFGTGLWVAVIM
TMRYSLKVLLSYHGWMFAEHGKMSRSTKIWMAMVKVLSGRKPMLYSFQTSLPRLPVPAVK
DTVSRYLESVRPLMKEEDFQRMTALAQDFAVNLGPKLQWYLKLKSWWATNYVSDWWEEYI
YLRGRGPLMVNSNYYAMEMLYITPTHIQAARAGNTIHAILLYRRTLDREELKPIRLLGST
IPLCSAQWERLFNTSRIPGEETDTIQHIKDSRHIVVYHRGRYFKVWLYHDGRLLRPRELE
QQMQQILDDPSEPQPGEAKLAALTAADRVPWAKCRQTYFARGKNKQSLDAVEKAAFFVTL
DESEQGYREEDPEASIDSYAKSLLHGRCFDRWFDKSITFVVFKNSKIGINAEHSWADAPV
VGHLWEYVMATDVFQLGYSEDGHCKGDTNPNIPKPTRLQWDIPGECQEVIDASLSSASLL
ANDVDLHSFPFDSFGKGLIKKCRTSPDAFIQLALQLAHYKDMGKFCLTYEASMTRLFREG
RTETVRSCTMESCNFVQAMMDPKSTAEQRLKLFKIACEKHQHLYRLAMTGAGIDRHLFCL
YVVSKYLAVDSPFLKEVLSEPWRLSTSQTPQQQVELFDFEKNPDYVSCGGGFGPVADDGY
GVSYIIVGENFIHFHISSKFSSPETDSHRFGKHLRQAMMDIITLFGLTINSKK
|
|
|
|---|
| BDBM50024430 |
|---|
| n/a |
|---|
| Name | BDBM50024430 |
|---|
| Synonyms: | (R) (R) -2-Tetradecyl-oxirane-2-carboxylic acid-Coenzyme A | CHEMBL12070 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C38H66N7O18P3S |
|---|
| Mol. Mass. | 1033.953 |
|---|
| SMILES | CCCCCCCCCCCCCC[C@@]1(CO1)C(=O)SCCNC(=O)CCNC(=O)C(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)n1cnc2c(N)ncnc12 |
|---|
| Structure | 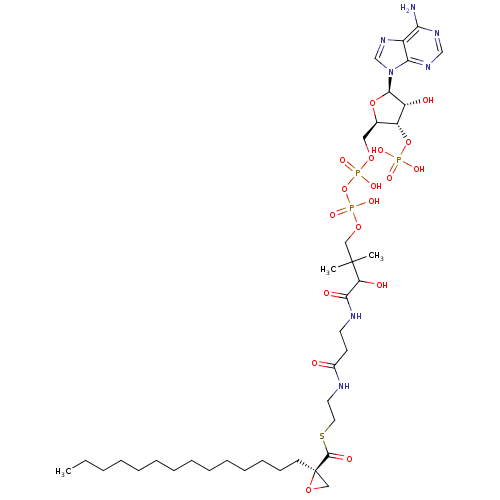
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ho, W; Tarhan, O; Kiorpes, TC; Tutwiler, GF; Mohrbacher, RJ Resolution of (+/-)-2-tetradecyloxiranecarboxylic acid. Absolute configuration and chiral synthesis of the hypoglycemic R enantiomer and biological activity of enantiomers. J Med Chem30:1094-7 (1987) [PubMed]
Ho, W; Tarhan, O; Kiorpes, TC; Tutwiler, GF; Mohrbacher, RJ Resolution of (+/-)-2-tetradecyloxiranecarboxylic acid. Absolute configuration and chiral synthesis of the hypoglycemic R enantiomer and biological activity of enantiomers. J Med Chem30:1094-7 (1987) [PubMed]