| Reaction Details |
|---|
 | Report a problem with these data |
| Target | DNA gyrase subunit B |
|---|
| Ligand | BDBM50226181 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1779332 (CHEMBL4236324) |
|---|
| IC50 | 20±n/a nM |
|---|
| Citation |  McGarry, DH; Cooper, IR; Walker, R; Warrilow, CE; Pichowicz, M; Ratcliffe, AJ; Salisbury, AM; Savage, VJ; Moyo, E; Maclean, J; Smith, A; Charrier, C; Stokes, NR; Lindsay, DM; Kerr, WJ Design, synthesis and antibacterial properties of pyrimido[4,5-b]indol-8-amine inhibitors of DNA gyrase. Bioorg Med Chem Lett28:2998-3003 (2018) [PubMed] Article McGarry, DH; Cooper, IR; Walker, R; Warrilow, CE; Pichowicz, M; Ratcliffe, AJ; Salisbury, AM; Savage, VJ; Moyo, E; Maclean, J; Smith, A; Charrier, C; Stokes, NR; Lindsay, DM; Kerr, WJ Design, synthesis and antibacterial properties of pyrimido[4,5-b]indol-8-amine inhibitors of DNA gyrase. Bioorg Med Chem Lett28:2998-3003 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| DNA gyrase subunit B |
|---|
| Name: | DNA gyrase subunit B |
|---|
| Synonyms: | 5.6.2.2 | DNA gyrase subunit B | GYRB_MYCTU | gyrB |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 74087.83 |
|---|
| Organism: | Mycobacterium tuberculosis (strain ATCC 25618 / H37Rv) |
|---|
| Description: | ChEMBL_109655 |
|---|
| Residue: | 675 |
|---|
| Sequence: | MAAQKKKAQDEYGAASITILEGLEAVRKRPGMYIGSTGERGLHHLIWEVVDNAVDEAMAG
YATTVNVVLLEDGGVEVADDGRGIPVATHASGIPTVDVVMTQLHAGGKFDSDAYAISGGL
HGVGVSVVNALSTRLEVEIKRDGYEWSQVYEKSEPLGLKQGAPTKKTGSTVRFWADPAVF
ETTEYDFETVARRLQEMAFLNKGLTINLTDERVTQDEVVDEVVSDVAEAPKSASERAAES
TAPHKVKSRTFHYPGGLVDFVKHINRTKNAIHSSIVDFSGKGTGHEVEIAMQWNAGYSES
VHTFANTINTHEGGTHEEGFRSALTSVVNKYAKDRKLLKDKDPNLTGDDIREGLAAVISV
KVSEPQFEGQTKTKLGNTEVKSFVQKVCNEQLTHWFEANPTDAKVVVNKAVSSAQARIAA
RKARELVRRKSATDIGGLPGKLADCRSTDPRKSELYVVEGDSAGGSAKSGRDSMFQAILP
LRGKIINVEKARIDRVLKNTEVQAIITALGTGIHDEFDIGKLRYHKIVLMADADVDGQHI
STLLLTLLFRFMRPLIENGHVFLAQPPLYKLKWQRSDPEFAYSDRERDGLLEAGLKAGKK
INKEDGIQRYKGLGEMDAKELWETTMDPSVRVLRQVTLDDAAAADELFSILMGEDVDARR
SFITRNAKDVRFLDV
|
|
|
|---|
| BDBM50226181 |
|---|
| n/a |
|---|
| Name | BDBM50226181 |
|---|
| Synonyms: | (3R,4S,5R,6R)-5-hydroxy-6-(4-hydroxy-3-(4-hydroxy-3-(3-methylbut-2-enyl)benzamido)-2-oxo-2H-chromen-7-yloxy)-3-methoxy-2,2-dimethyl-tetrahydro-2H-pyran-4-yl carbamate | (3R,4S,5R,6R)-5-hydroxy-6-(4-hydroxy-3-(4-hydroxy-3-(3-methylbut-2-enyl)benzamido)-8-methyl-2-oxo-2H-chromen-7-yloxy)-3-methoxy-2,2-dimethyl-tetrahydro-2H-pyran-4-yl carbamate | CHEMBL36506 | Carbamic acid (3R,4S,5R,6R)-5-hydroxy-6-{4-hydroxy-3-[4-hydroxy-3-(3-methyl-but-2-enyl)-benzoylamino]-8-methyl-2-oxo-2H-chromen-7-yloxy}-3-methoxy-2,2-dimethyl-tetrahydro-pyran-4-yl ester | Carbamic acid (3R,4S,5R,6R)-5-hydroxy-6-{4-hydroxy-3-[4-hydroxy-3-(4-methyl-pent-3-enyl)-benzoylamino]-8-methyl-2-oxo-2H-chromen-7-yloxy}-3-methoxy-2,2-dimethyl-tetrahydro-pyran-4-yl ester | Carbamic acid 5-hydroxy-6-{4-hydroxy-3-[4-hydroxy-3-(3-methyl-but-2-enyl)-benzoylamino]-8-methyl-2-oxo-2H-chromen-7-yloxy}-3-methoxy-2,2-dimethyl-tetrahydro-pyran-4-yl ester | Carbamic acid 5-hydroxy-6-{4-hydroxy-3-[4-hydroxy-3-(3-methyl-but-2-enyl)-benzoylamino]-8-methyl-2-oxo-2H-chromen-7-yloxy}-3-methoxy-2,2-dimethyl-tetrahydro-pyran-4-yl ester(Novobiocin) | NOVOBIOCIN |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H36N2O11 |
|---|
| Mol. Mass. | 612.6243 |
|---|
| SMILES | [#6]-[#8]-[#6@@H]1-[#6@@H](-[#8]-[#6](-[#7])=O)-[#6@@H](-[#8])-[#6@H](-[#8]-c2ccc3c(-[#8])c(-[#7]-[#6](=O)-c4ccc(-[#8])c(-[#6]\[#6]=[#6](/[#6])-[#6])c4)c(=O)oc3c2-[#6])-[#8]C1([#6])[#6] |r| |
|---|
| Structure | 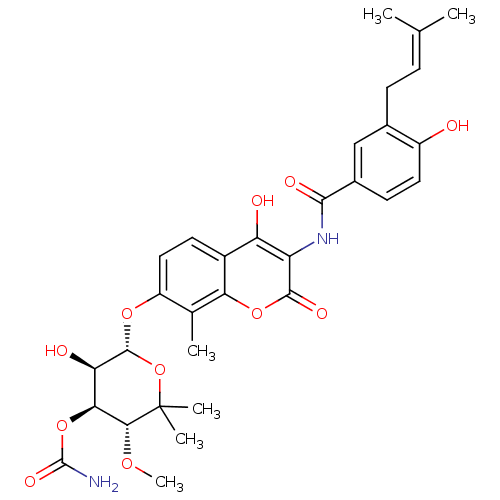
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 McGarry, DH; Cooper, IR; Walker, R; Warrilow, CE; Pichowicz, M; Ratcliffe, AJ; Salisbury, AM; Savage, VJ; Moyo, E; Maclean, J; Smith, A; Charrier, C; Stokes, NR; Lindsay, DM; Kerr, WJ Design, synthesis and antibacterial properties of pyrimido[4,5-b]indol-8-amine inhibitors of DNA gyrase. Bioorg Med Chem Lett28:2998-3003 (2018) [PubMed] Article
McGarry, DH; Cooper, IR; Walker, R; Warrilow, CE; Pichowicz, M; Ratcliffe, AJ; Salisbury, AM; Savage, VJ; Moyo, E; Maclean, J; Smith, A; Charrier, C; Stokes, NR; Lindsay, DM; Kerr, WJ Design, synthesis and antibacterial properties of pyrimido[4,5-b]indol-8-amine inhibitors of DNA gyrase. Bioorg Med Chem Lett28:2998-3003 (2018) [PubMed] Article