| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histamine H4 receptor |
|---|
| Ligand | BDBM50021529 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1799139 (CHEMBL4271431) |
|---|
| Ki | 520±n/a nM |
|---|
| Citation |  Cascioferro, S; Parrino, B; Span�, V; Carbone, A; Montalbano, A; Barraja, P; Diana, P; Cirrincione, G 1,3,5-Triazines: A promising scaffold for anticancer drugs development. Eur J Med Chem142:523-549 (2017) [PubMed] Article Cascioferro, S; Parrino, B; Span�, V; Carbone, A; Montalbano, A; Barraja, P; Diana, P; Cirrincione, G 1,3,5-Triazines: A promising scaffold for anticancer drugs development. Eur J Med Chem142:523-549 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histamine H4 receptor |
|---|
| Name: | Histamine H4 receptor |
|---|
| Synonyms: | AXOR35 | G-protein coupled receptor 105 | GPCR105 | GPRv53 | HH4R | HISTAMINE H4 | HRH4 | HRH4_HUMAN | Histamine H4 receptor | Histamine H4 receptor (H4R) | Histamine receptor (H3 and H4) | Pfi-013 | SP9144 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44517.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Binding assays were using CHO cells stably expressing hH4R receptors. |
|---|
| Residue: | 390 |
|---|
| Sequence: | MPDTNSTINLSLSTRVTLAFFMSLVAFAIMLGNALVILAFVVDKNLRHRSSYFFLNLAIS
DFFVGVISIPLYIPHTLFEWDFGKEICVFWLTTDYLLCTASVYNIVLISYDRYLSVSNAV
SYRTQHTGVLKIVTLMVAVWVLAFLVNGPMILVSESWKDEGSECEPGFFSEWYILAITSF
LEFVIPVILVAYFNMNIYWSLWKRDHLSRCQSHPGLTAVSSNICGHSFRGRLSSRRSLSA
STEVPASFHSERQRRKSSLMFSSRTKMNSNTIASKMGSFSQSDSVALHQREHVELLRARR
LAKSLAILLGVFAVCWAPYSLFTIVLSFYSSATGPKSVWYRIAFWLQWFNSFVNPLLYPL
CHKRFQKAFLKIFCIKKQPLPSQHSRSVSS
|
|
|
|---|
| BDBM50021529 |
|---|
| n/a |
|---|
| Name | BDBM50021529 |
|---|
| Synonyms: | CHEMBL3290578 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H17BrN6 |
|---|
| Mol. Mass. | 349.229 |
|---|
| SMILES | CN1CCN(CC1)c1nc(N)nc(n1)-c1ccc(Br)cc1 |
|---|
| Structure | 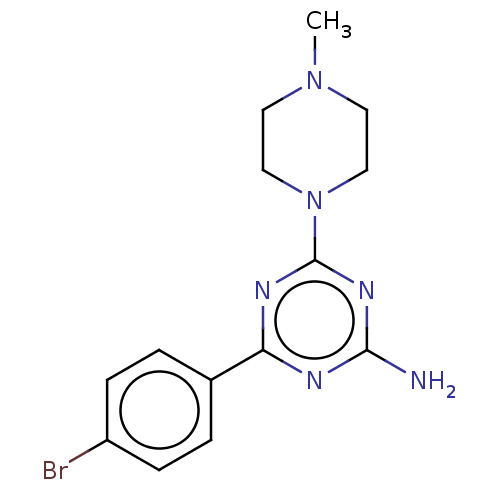
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cascioferro, S; Parrino, B; Span�, V; Carbone, A; Montalbano, A; Barraja, P; Diana, P; Cirrincione, G 1,3,5-Triazines: A promising scaffold for anticancer drugs development. Eur J Med Chem142:523-549 (2017) [PubMed] Article
Cascioferro, S; Parrino, B; Span�, V; Carbone, A; Montalbano, A; Barraja, P; Diana, P; Cirrincione, G 1,3,5-Triazines: A promising scaffold for anticancer drugs development. Eur J Med Chem142:523-549 (2017) [PubMed] Article