| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-2A adrenergic receptor |
|---|
| Ligand | BDBM30712 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_33200 (CHEMBL640501) |
|---|
| Ki | 16±n/a nM |
|---|
| Citation |  Hodson, SJ; Bishop, MJ; Speake, JD; Navas, F; Garrison, DT; Bigham, EC; Saussy, DL; Liacos, JA; Irving, PE; Gobel, MJ; Sherman, BW 2-(Anilinomethyl)imidazolines as alpha1 adrenergic receptor agonists: the discovery of alpha1a subtype selective 2'-alkylsulfonyl-substituted analogues. J Med Chem45:2229-39 (2002) [PubMed] Article Hodson, SJ; Bishop, MJ; Speake, JD; Navas, F; Garrison, DT; Bigham, EC; Saussy, DL; Liacos, JA; Irving, PE; Gobel, MJ; Sherman, BW 2-(Anilinomethyl)imidazolines as alpha1 adrenergic receptor agonists: the discovery of alpha1a subtype selective 2'-alkylsulfonyl-substituted analogues. J Med Chem45:2229-39 (2002) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-2A adrenergic receptor |
|---|
| Name: | Alpha-2A adrenergic receptor |
|---|
| Synonyms: | ADA2A_HUMAN | ADRA2A | ADRA2R | ADRAR | Adrenergic alpha2A | Adrenergic receptor alpha | Alpha-2 adrenergic receptor subtype C10 | Alpha-2A adrenoceptor | Alpha-2A adrenoreceptor | Alpha-2AAR | alpha-2A adrenergic receptor [Homo sapiens] |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48979.91 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P08913 |
|---|
| Residue: | 465 |
|---|
| Sequence: | MFRQEQPLAEGSFAPMGSLQPDAGNASWNGTEAPGGGARATPYSLQVTLTLVCLAGLLML
LTVFGNVLVIIAVFTSRALKAPQNLFLVSLASADILVATLVIPFSLANEVMGYWYFGKAW
CEIYLALDVLFCTSSIVHLCAISLDRYWSITQAIEYNLKRTPRRIKAIIITVWVISAVIS
FPPLISIEKKGGGGGPQPAEPRCEINDQKWYVISSCIGSFFAPCLIMILVYVRIYQIAKR
RTRVPPSRRGPDAVAAPPGGTERRPNGLGPERSAGPGGAEAEPLPTQLNGAPGEPAPAGP
RDTDALDLEESSSSDHAERPPGPRRPERGPRGKGKARASQVKPGDSLPRRGPGATGIGTP
AAGPGEERVGAAKASRWRGRQNREKRFTFVLAVVIGVFVVCWFPFFFTYTLTAVGCSVPR
TLFKFFFWFGYCNSSLNPVIYTIFNHDFRRAFKKILCRGDRKRIV
|
|
|
|---|
| BDBM30712 |
|---|
| n/a |
|---|
| Name | BDBM30712 |
|---|
| Synonyms: | 6-tert-butyl-3-(2-imidazolin-2-ylmethyl)-2,4-dimethyl-phenol;hydrochloride | 6-tert-butyl-3-(4,5-dihydro-1H-imidazol-2-ylmethyl)-2,4-dimethyl-phenol;hydrochloride | 6-tert-butyl-3-(4,5-dihydro-1H-imidazol-2-ylmethyl)-2,4-dimethylphenol;hydrochloride | MLS000038040 | Oxymetazoline | SMR000059324 | cid_66259 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H24N2O |
|---|
| Mol. Mass. | 260.3746 |
|---|
| SMILES | Cc1cc(c(O)c(C)c1CC1=NCCN1)C(C)(C)C |t:11| |
|---|
| Structure | 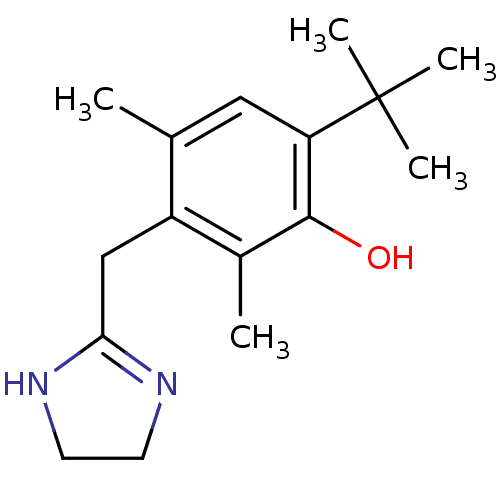
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hodson, SJ; Bishop, MJ; Speake, JD; Navas, F; Garrison, DT; Bigham, EC; Saussy, DL; Liacos, JA; Irving, PE; Gobel, MJ; Sherman, BW 2-(Anilinomethyl)imidazolines as alpha1 adrenergic receptor agonists: the discovery of alpha1a subtype selective 2'-alkylsulfonyl-substituted analogues. J Med Chem45:2229-39 (2002) [PubMed] Article
Hodson, SJ; Bishop, MJ; Speake, JD; Navas, F; Garrison, DT; Bigham, EC; Saussy, DL; Liacos, JA; Irving, PE; Gobel, MJ; Sherman, BW 2-(Anilinomethyl)imidazolines as alpha1 adrenergic receptor agonists: the discovery of alpha1a subtype selective 2'-alkylsulfonyl-substituted analogues. J Med Chem45:2229-39 (2002) [PubMed] Article