| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Penicillin-binding protein 1A |
|---|
| Ligand | BDBM50482777 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_672067 (CHEMBL1267448) |
|---|
| IC50 | 219±n/a nM |
|---|
| Citation |  Kosowska-Shick, K; McGhee, PL; Appelbaum, PC Affinity of ceftaroline and other beta-lactams for penicillin-binding proteins from Staphylococcus aureus and Streptococcus pneumoniae. Antimicrob Agents Chemother54:1670-7 (2010) [PubMed] Article Kosowska-Shick, K; McGhee, PL; Appelbaum, PC Affinity of ceftaroline and other beta-lactams for penicillin-binding proteins from Staphylococcus aureus and Streptococcus pneumoniae. Antimicrob Agents Chemother54:1670-7 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Penicillin-binding protein 1A |
|---|
| Name: | Penicillin-binding protein 1A |
|---|
| Synonyms: | 2.4.1.129 | 3.4.16.4 | DD-transpeptidase | PBP-1A | PBPA_STRPN | Penicillin-binding protein 1A | Penicillin-insensitive transglycosylase | Penicillin-sensitive transpeptidase | Peptidoglycan TGase | ponA |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 79749.99 |
|---|
| Organism: | Streptococcus pneumoniae serotype 4 (strain ATCC BAA-334 / TIGR4) |
|---|
| Description: | ChEMBL_103552 |
|---|
| Residue: | 719 |
|---|
| Sequence: | MNKPTILRLIKYLSISFLSLVIAAIVLGGGVFFYYVSKAPSLSESKLVATTSSKIYDNKN
QLIADLGSERRVNAQANDIPTDLVKAIVSIEDHRFFDHRGIDTIRILGAFLRNLQSNSLQ
GGSTLTQQLIKLTYFSTSTSDQTISRKAQEAWLAIQLEQKATKQEILTYYINKVYMSNGN
YGMQTAAQNYYGKDLNNLSLPQLALLAGMPQAPNQYDPYSHPEAAQDRRNLVLSEMKNQG
YISAEQYEKAVNTPITDGLQSLKSASNYPAYMDNYLKEVINQVEEETGYNLLTTGMDVYT
NVDQEAQKHLWDIYNTDEYVAYPDDELQVASTIVDVSNGKVIAQLGARHQSSNVSFGINQ
AVETNRDWGSTMKPITDYAPALEYGVYDSTATIVHDEPYNYPGTNTPVYNWDRGYFGNIT
LQYALQQSRNVPAVETLNKVGLNRAKTFLNGLGIDYPSIHYSNAISSNTTESDKKYGASS
EKMAAAYAAFANGGTYYKPMYIHKVVFSDGSEKEFSNVGTRAMKETTAYMMTDMMKTVLT
YGTGRNAYLAWLPQAGKTGTSNYTDEEIENHIKTSQFVAPDELFAGYTRKYSMAVWTGYS
NRLTPLVGNGLTVAAKVYRSMMTYLSEGSNPEDWNIPEGLYRNGEFVFKNGARSTWNSPA
PQQPPSTESSSSSSDSSTSQSSSTTPSTNNSTTTNPNNNTQQSNTTPDQQNQNPQPAQP
|
|
|
|---|
| BDBM50482777 |
|---|
| n/a |
|---|
| Name | BDBM50482777 |
|---|
| Synonyms: | CHEBI:204928 | Cefotaxime |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H17N5O7S2 |
|---|
| Mol. Mass. | 455.465 |
|---|
| SMILES | [H][C@]12SCC(COC(C)=O)=C(N1C(=O)[C@H]2NC(=O)C(=N/OC)\c1csc(N)n1)C(O)=O |c:9| |
|---|
| Structure | 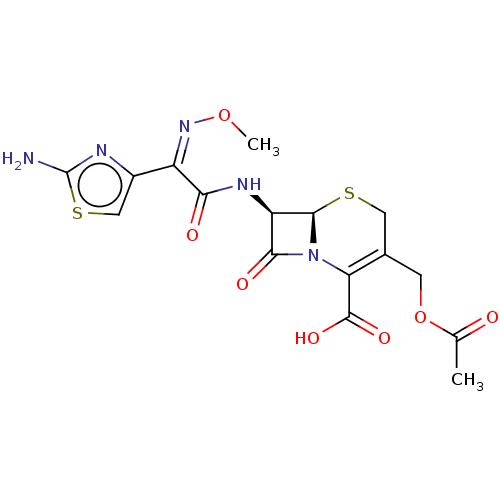
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kosowska-Shick, K; McGhee, PL; Appelbaum, PC Affinity of ceftaroline and other beta-lactams for penicillin-binding proteins from Staphylococcus aureus and Streptococcus pneumoniae. Antimicrob Agents Chemother54:1670-7 (2010) [PubMed] Article
Kosowska-Shick, K; McGhee, PL; Appelbaum, PC Affinity of ceftaroline and other beta-lactams for penicillin-binding proteins from Staphylococcus aureus and Streptococcus pneumoniae. Antimicrob Agents Chemother54:1670-7 (2010) [PubMed] Article