| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Aromatase |
|---|
| Ligand | BDBM50044424 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_51346 |
|---|
| Ki | 7.6±n/a nM |
|---|
| Citation |  Walker, KA; Kertesz, DJ; Rotstein, DM; Swinney, DC; Berry, PW; So, OY; Webb, AS; Watson, DM; Mak, AY; Burton, PM Selective inhibition of mammalian lanosterol 14 alpha-demethylase: a possible strategy for cholesterol lowering. J Med Chem36:2235-7 (1993) [PubMed] Walker, KA; Kertesz, DJ; Rotstein, DM; Swinney, DC; Berry, PW; So, OY; Webb, AS; Watson, DM; Mak, AY; Burton, PM Selective inhibition of mammalian lanosterol 14 alpha-demethylase: a possible strategy for cholesterol lowering. J Med Chem36:2235-7 (1993) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Aromatase |
|---|
| Name: | Aromatase |
|---|
| Synonyms: | ARO1 | Aromatase | CP19A_HUMAN | CYAR | CYP19 | CYP19A1 | CYPXIX | Cytochrome P-450AROM | Cytochrome P450 19A1 | Cytochrome P450 2C19 | Cytochrome P450-C19 (CYP19) | Estrogen synthetase | FL cytokine receptor precursor | P-450AROM |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57888.92 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11511 |
|---|
| Residue: | 503 |
|---|
| Sequence: | MVLEMLNPIHYNITSIVPEAMPAATMPVLLLTGLFLLVWNYEGTSSIPGPGYCMGIGPLI
SHGRFLWMGIGSACNYYNRVYGEFMRVWISGEETLIISKSSSMFHIMKHNHYSSRFGSKL
GLQCIGMHEKGIIFNNNPELWKTTRPFFMKALSGPGLVRMVTVCAESLKTHLDRLEEVTN
ESGYVDVLTLLRRVMLDTSNTLFLRIPLDESAIVVKIQGYFDAWQALLIKPDIFFKISWL
YKKYEKSVKDLKDAIEVLIAEKRRRISTEEKLEECMDFATELILAEKRGDLTRENVNQCI
LEMLIAAPDTMSVSLFFMLFLIAKHPNVEEAIIKEIQTVIGERDIKIDDIQKLKVMENFI
YESMRYQPVVDLVMRKALEDDVIDGYPVKKGTNIILNIGRMHRLEFFPKPNEFTLENFAK
NVPYRYFQPFGFGPRGCAGKYIAMVMMKAILVTLLRRFHVKTLQGQCVESIQKIHDLSLH
PDETKNMLEMIFTPRNSDRCLEH
|
|
|
|---|
| BDBM50044424 |
|---|
| n/a |
|---|
| Name | BDBM50044424 |
|---|
| Synonyms: | 4-(((2S,4S)-2-((1H-imidazol-1-yl)methyl)-2-(4-chlorophenethyl)-1,3-dioxolan-4-yl)methylthio)benzenamine | 4-{(2S,4S)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazol-1-ylmethyl-[1,3]dioxolan-4-ylmethylsulfanyl}-phenylamine | 4-{2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazol-1-ylmethyl-[1,3]dioxolan-4-ylmethylsulfanyl}-phenylamine | Azalanstat dihydrochloride | CHEMBL70611 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H24ClN3O2S |
|---|
| Mol. Mass. | 429.963 |
|---|
| SMILES | Nc1ccc(SC[C@@H]2CO[C@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 |
|---|
| Structure | 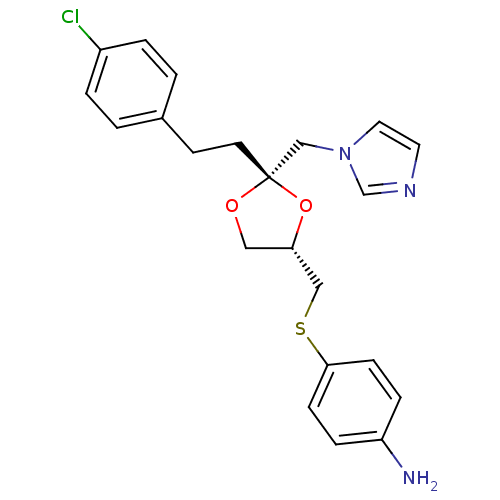
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Walker, KA; Kertesz, DJ; Rotstein, DM; Swinney, DC; Berry, PW; So, OY; Webb, AS; Watson, DM; Mak, AY; Burton, PM Selective inhibition of mammalian lanosterol 14 alpha-demethylase: a possible strategy for cholesterol lowering. J Med Chem36:2235-7 (1993) [PubMed]
Walker, KA; Kertesz, DJ; Rotstein, DM; Swinney, DC; Berry, PW; So, OY; Webb, AS; Watson, DM; Mak, AY; Burton, PM Selective inhibition of mammalian lanosterol 14 alpha-demethylase: a possible strategy for cholesterol lowering. J Med Chem36:2235-7 (1993) [PubMed]