| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glutaminase kidney isoform, mitochondrial |
|---|
| Ligand | BDBM332752 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1809975 (CHEMBL4309435) |
|---|
| IC50 | 25±n/a nM |
|---|
| Citation |  Zimmermann, SC; Duvall, B; Tsukamoto, T Recent Progress in the Discovery of Allosteric Inhibitors of Kidney-Type Glutaminase. J Med Chem62:46-59 (2019) [PubMed] Article Zimmermann, SC; Duvall, B; Tsukamoto, T Recent Progress in the Discovery of Allosteric Inhibitors of Kidney-Type Glutaminase. J Med Chem62:46-59 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glutaminase kidney isoform, mitochondrial |
|---|
| Name: | Glutaminase kidney isoform, mitochondrial |
|---|
| Synonyms: | GLS | GLS1 | GLSK_HUMAN | Glutaminase 1 | K-glutaminase | KIAA0838 | L-glutamine amidohydrolase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 73471.89 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O94925 |
|---|
| Residue: | 669 |
|---|
| Sequence: | MMRLRGSGMLRDLLLRSPAGVSATLRRAQPLVTLCRRPRGGGRPAAGPAAAARLHPWWGG
GGWPAEPLARGLSSSPSEILQELGKGSTHPQPGVSPPAAPAAPGPKDGPGETDAFGNSEG
KELVASGENKIKQGLLPSLEDLLFYTIAEGQEKIPVHKFITALKSTGLRTSDPRLKECMD
MLRLTLQTTSDGVMLDKDLFKKCVQSNIVLLTQAFRRKFVIPDFMSFTSHIDELYESAKK
QSGGKVADYIPQLAKFSPDLWGVSVCTVDGQRHSTGDTKVPFCLQSCVKPLKYAIAVNDL
GTEYVHRYVGKEPSGLRFNKLFLNEDDKPHNPMVNAGAIVVTSLIKQGVNNAEKFDYVMQ
FLNKMAGNEYVGFSNATFQSERESGDRNFAIGYYLKEKKCFPEGTDMVGILDFYFQLCSI
EVTCESASVMAATLANGGFCPITGERVLSPEAVRNTLSLMHSCGMYDFSGQFAFHVGLPA
KSGVAGGILLVVPNVMGMMCWSPPLDKMGNSVKGIHFCHDLVSLCNFHNYDNLRHFAKKL
DPRREGGDQRVKSVINLLFAAYTGDVSALRRFALSAMDMEQRDYDSRTALHVAAAEGHVE
VVKFLLEACKVNPFPKDRWNNTPMDEALHFGHHDVFKILQEYQVQYTPQGDSDNGKENQT
VHKNLDGLL
|
|
|
|---|
| BDBM332752 |
|---|
| n/a |
|---|
| Name | BDBM332752 |
|---|
| Synonyms: | (2S)-2-[3-(Difluoromethoxy)phenyl]-2-methoxy-N-[6-[(3R)-3-(pyridazin-3-ylamino)pyrrolidin-1-yl]pyridazin-3-yl]acetamide | US10196382, Example 1 | US10577354, Example 1 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H23F2N7O3 |
|---|
| Mol. Mass. | 471.4599 |
|---|
| SMILES | CO[C@H](C(=O)Nc1ccc(nn1)N1CC[C@H](C1)Nc1cccnn1)c1cccc(OC(F)F)c1 |r| |
|---|
| Structure | 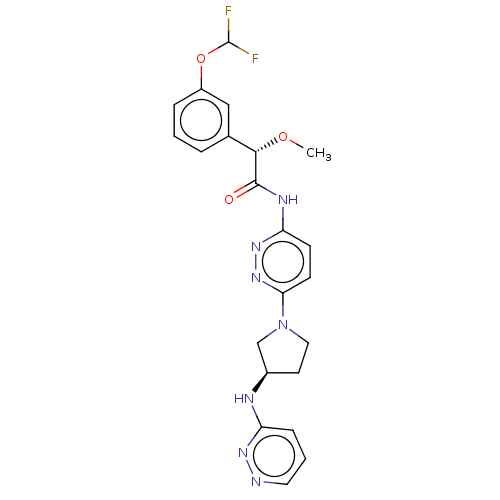
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zimmermann, SC; Duvall, B; Tsukamoto, T Recent Progress in the Discovery of Allosteric Inhibitors of Kidney-Type Glutaminase. J Med Chem62:46-59 (2019) [PubMed] Article
Zimmermann, SC; Duvall, B; Tsukamoto, T Recent Progress in the Discovery of Allosteric Inhibitors of Kidney-Type Glutaminase. J Med Chem62:46-59 (2019) [PubMed] Article