| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Muscarinic acetylcholine receptor M2 |
|---|
| Ligand | BDBM46858 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_138184 (CHEMBL749205) |
|---|
| IC50 | 4500±n/a nM |
|---|
| Citation |  Sauerberg, P; Olesen, PH; Sheardown, MJ; Rimvall, K; Thøgersen, H; Shannon, HE; Sawyer, BD; Ward, JS; Bymaster, FP; DeLapp, NW; Calligaro, DO; Swedberg, MD Conformationally constrained analogues of the muscarinic agonist 3-(4-(methylthio)-1,2,5-thiadiazol-3-yl)-1,2,5,6-tetrahydro-1-methylpyr idine. Synthesis, receptor affinity, and antinociceptive activity. J Med Chem41:109-16 (1998) [PubMed] Article Sauerberg, P; Olesen, PH; Sheardown, MJ; Rimvall, K; Thøgersen, H; Shannon, HE; Sawyer, BD; Ward, JS; Bymaster, FP; DeLapp, NW; Calligaro, DO; Swedberg, MD Conformationally constrained analogues of the muscarinic agonist 3-(4-(methylthio)-1,2,5-thiadiazol-3-yl)-1,2,5,6-tetrahydro-1-methylpyr idine. Synthesis, receptor affinity, and antinociceptive activity. J Med Chem41:109-16 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Muscarinic acetylcholine receptor M2 |
|---|
| Name: | Muscarinic acetylcholine receptor M2 |
|---|
| Synonyms: | ACM2_HUMAN | CHRM2 | Cholinergic, muscarinic M2 | Muscarinic acetylcholine receptor M2 and M4 | Muscarinic acetylcholine receptor M2 and M5 | RecName: Full=Muscarinic acetylcholine receptor M2 |
|---|
| Type: | GPCR |
|---|
| Mol. Mass.: | 51730.61 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P08172 |
|---|
| Residue: | 466 |
|---|
| Sequence: | MNNSTNSSNNSLALTSPYKTFEVVFIVLVAGSLSLVTIIGNILVMVSIKVNRHLQTVNNY
FLFSLACADLIIGVFSMNLYTLYTVIGYWPLGPVVCDLWLALDYVVSNASVMNLLIISFD
RYFCVTKPLTYPVKRTTKMAGMMIAAAWVLSFILWAPAILFWQFIVGVRTVEDGECYIQF
FSNAAVTFGTAIAAFYLPVIIMTVLYWHISRASKSRIKKDKKEPVANQDPVSPSLVQGRI
VKPNNNNMPSSDDGLEHNKIQNGKAPRDPVTENCVQGEEKESSNDSTSVSAVASNMRDDE
ITQDENTVSTSLGHSKDENSKQTCIRIGTKTPKSDSCTPTNTTVEVVGSSGQNGDEKQNI
VARKIVKMTKQPAKKKPPPSREKKVTRTILAILLAFIITWAPYNVMVLINTFCAPCIPNT
VWTIGYWLCYINSTINPACYALCNATFKKTFKHLLMCHYKNIGATR
|
|
|
|---|
| BDBM46858 |
|---|
| n/a |
|---|
| Name | BDBM46858 |
|---|
| Synonyms: | 1-methyl-3,6-dihydro-2H-pyridine-5-carboxylic acid methyl ester;hydrobromide | ARECOLINE | ARECOLINE HYDROBROMIDE | MLS000028840 | SMR000058258 | arecoline;hydrobromide | cid_9301 | methyl 1-methyl-3,6-dihydro-2H-pyridine-5-carboxylate;hydrobromide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C8H13NO2 |
|---|
| Mol. Mass. | 155.1943 |
|---|
| SMILES | COC(=O)C1=CCCN(C)C1 |t:4| |
|---|
| Structure | 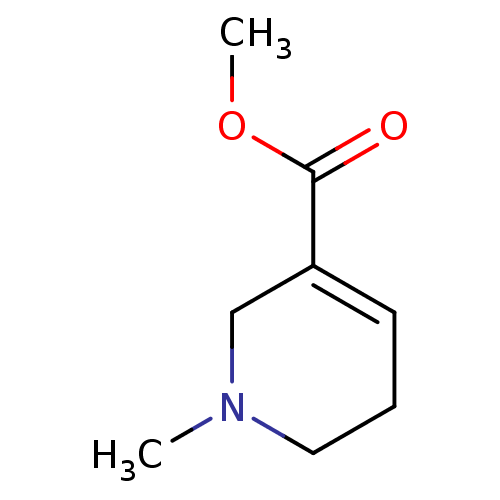
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sauerberg, P; Olesen, PH; Sheardown, MJ; Rimvall, K; Thøgersen, H; Shannon, HE; Sawyer, BD; Ward, JS; Bymaster, FP; DeLapp, NW; Calligaro, DO; Swedberg, MD Conformationally constrained analogues of the muscarinic agonist 3-(4-(methylthio)-1,2,5-thiadiazol-3-yl)-1,2,5,6-tetrahydro-1-methylpyr idine. Synthesis, receptor affinity, and antinociceptive activity. J Med Chem41:109-16 (1998) [PubMed] Article
Sauerberg, P; Olesen, PH; Sheardown, MJ; Rimvall, K; Thøgersen, H; Shannon, HE; Sawyer, BD; Ward, JS; Bymaster, FP; DeLapp, NW; Calligaro, DO; Swedberg, MD Conformationally constrained analogues of the muscarinic agonist 3-(4-(methylthio)-1,2,5-thiadiazol-3-yl)-1,2,5,6-tetrahydro-1-methylpyr idine. Synthesis, receptor affinity, and antinociceptive activity. J Med Chem41:109-16 (1998) [PubMed] Article