| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2b |
|---|
| Ligand | BDBM50053923 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_30295 (CHEMBL875505) |
|---|
| IC50 | 2650±n/a nM |
|---|
| Citation |  Kim, YC; de Zwart, M; Chang, L; Moro, S; von Frijtag Drabbe Künzel, JK; Melman, N; IJzerman, AP; Jacobson, KA Derivatives of the triazoloquinazoline adenosine antagonist (CGS 15943) having high potency at the human A2B and A3 receptor subtypes. J Med Chem41:2835-45 (1998) [PubMed] Article Kim, YC; de Zwart, M; Chang, L; Moro, S; von Frijtag Drabbe Künzel, JK; Melman, N; IJzerman, AP; Jacobson, KA Derivatives of the triazoloquinazoline adenosine antagonist (CGS 15943) having high potency at the human A2B and A3 receptor subtypes. J Med Chem41:2835-45 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2b |
|---|
| Name: | Adenosine receptor A2b |
|---|
| Synonyms: | AA2BR_HUMAN | ADENOSINE A2B | ADORA2B | Adenosine receptor A2B (A2B) | Adenosine receptors A2b | Adenosine receptors; A2a & A2b |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 36341.22 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 332 |
|---|
| Sequence: | MLLETQDALYVALELVIAALSVAGNVLVCAAVGTANTLQTPTNYFLVSLAAADVAVGLFA
IPFAITISLGFCTDFYGCLFLACFVLVLTQSSIFSLLAVAVDRYLAICVPLRYKSLVTGT
RARGVIAVLWVLAFGIGLTPFLGWNSKDSATNNCTEPWDGTTNESCCLVKCLFENVVPMS
YMVYFNFFGCVLPPLLIMLVIYIKIFLVACRQLQRTELMDHSRTTLQREIHAAKSLAMIV
GIFALCWLPVHAVNCVTLFQPAQGKNKPKWAMNMAILLSHANSVVNPIVYAYRNRDFRYT
FHKIISRYLLCQADVKSGNGQAGVQPALGVGL
|
|
|
|---|
| BDBM50053923 |
|---|
| n/a |
|---|
| Name | BDBM50053923 |
|---|
| Synonyms: | CHEMBL318251 | N-(9-Chloro-2-furan-2-yl-[1,2,4]triazolo[1,5-c]quinazolin-5-yl)-acetamide | N-(9-chloro-2-(furan-2-yl)-[1,2,4]triazolo[1,5-c]quinazolin-5-yl)acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H10ClN5O2 |
|---|
| Mol. Mass. | 327.725 |
|---|
| SMILES | CC(=O)Nc1nc2ccc(Cl)cc2c2nc(nn12)-c1ccco1 |
|---|
| Structure | 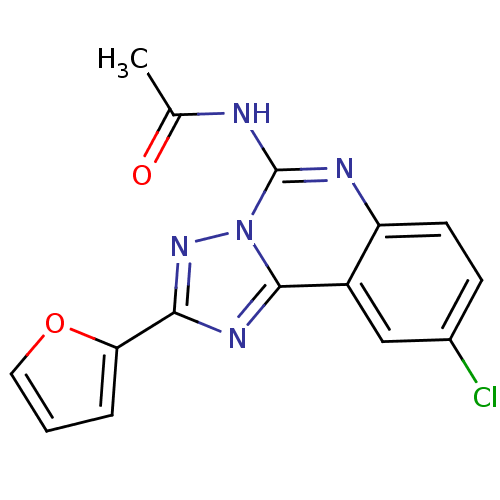
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kim, YC; de Zwart, M; Chang, L; Moro, S; von Frijtag Drabbe Künzel, JK; Melman, N; IJzerman, AP; Jacobson, KA Derivatives of the triazoloquinazoline adenosine antagonist (CGS 15943) having high potency at the human A2B and A3 receptor subtypes. J Med Chem41:2835-45 (1998) [PubMed] Article
Kim, YC; de Zwart, M; Chang, L; Moro, S; von Frijtag Drabbe Künzel, JK; Melman, N; IJzerman, AP; Jacobson, KA Derivatives of the triazoloquinazoline adenosine antagonist (CGS 15943) having high potency at the human A2B and A3 receptor subtypes. J Med Chem41:2835-45 (1998) [PubMed] Article