| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM50519778 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1878392 (CHEMBL4379786) |
|---|
| Ki | 5.2±n/a nM |
|---|
| Citation |  Garai, S; Kulkarni, PM; Schaffer, PC; Leo, LM; Brandt, AL; Zagzoog, A; Black, T; Lin, X; Hurst, DP; Janero, DR; Abood, ME; Zimmowitch, A; Straiker, A; Pertwee, RG; Kelly, M; Szczesniak, AM; Denovan-Wright, EM; Mackie, K; Hohmann, AG; Reggio, PH; Laprairie, RB; Thakur, GA Application of Fluorine- and Nitrogen-Walk Approaches: Defining the Structural and Functional Diversity of 2-Phenylindole Class of Cannabinoid 1 Receptor Positive Allosteric Modulators. J Med Chem63:542-568 (2020) [PubMed] Article Garai, S; Kulkarni, PM; Schaffer, PC; Leo, LM; Brandt, AL; Zagzoog, A; Black, T; Lin, X; Hurst, DP; Janero, DR; Abood, ME; Zimmowitch, A; Straiker, A; Pertwee, RG; Kelly, M; Szczesniak, AM; Denovan-Wright, EM; Mackie, K; Hohmann, AG; Reggio, PH; Laprairie, RB; Thakur, GA Application of Fluorine- and Nitrogen-Walk Approaches: Defining the Structural and Functional Diversity of 2-Phenylindole Class of Cannabinoid 1 Receptor Positive Allosteric Modulators. J Med Chem63:542-568 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM50519778 |
|---|
| n/a |
|---|
| Name | BDBM50519778 |
|---|
| Synonyms: | CHEMBL4519310 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H15F3N2O2 |
|---|
| Mol. Mass. | 396.3619 |
|---|
| SMILES | [O-][N+](=O)CC(c1c([nH]c2c(F)cccc12)-c1cccc(F)c1)c1ccccc1F |
|---|
| Structure | 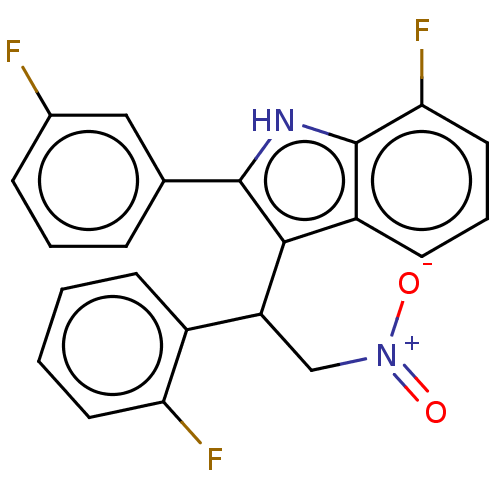
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Garai, S; Kulkarni, PM; Schaffer, PC; Leo, LM; Brandt, AL; Zagzoog, A; Black, T; Lin, X; Hurst, DP; Janero, DR; Abood, ME; Zimmowitch, A; Straiker, A; Pertwee, RG; Kelly, M; Szczesniak, AM; Denovan-Wright, EM; Mackie, K; Hohmann, AG; Reggio, PH; Laprairie, RB; Thakur, GA Application of Fluorine- and Nitrogen-Walk Approaches: Defining the Structural and Functional Diversity of 2-Phenylindole Class of Cannabinoid 1 Receptor Positive Allosteric Modulators. J Med Chem63:542-568 (2020) [PubMed] Article
Garai, S; Kulkarni, PM; Schaffer, PC; Leo, LM; Brandt, AL; Zagzoog, A; Black, T; Lin, X; Hurst, DP; Janero, DR; Abood, ME; Zimmowitch, A; Straiker, A; Pertwee, RG; Kelly, M; Szczesniak, AM; Denovan-Wright, EM; Mackie, K; Hohmann, AG; Reggio, PH; Laprairie, RB; Thakur, GA Application of Fluorine- and Nitrogen-Walk Approaches: Defining the Structural and Functional Diversity of 2-Phenylindole Class of Cannabinoid 1 Receptor Positive Allosteric Modulators. J Med Chem63:542-568 (2020) [PubMed] Article