

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Corticotropin-releasing factor receptor 1 | ||
| Ligand | BDBM50074455 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_51114 (CHEMBL664960) | ||
| Ki | 7.1±n/a nM | ||
| Citation |  Chorvat, RJ; Bakthavatchalam, R; Beck, JP; Gilligan, PJ; Wilde, RG; Cocuzza, AJ; Hobbs, FW; Cheeseman, RS; Curry, M; Rescinito, JP; Krenitsky, P; Chidester, D; Yarem, JA; Klaczkiewicz, JD; Hodge, CN; Aldrich, PE; Wasserman, ZR; Fernandez, CH; Zaczek, R; Fitzgerald, LW; Huang, SM; Shen, HL; Wong, YN; Chien, BM; Arvanitis, A Synthesis, corticotropin-releasing factor receptor binding affinity, and pharmacokinetic properties of triazolo-, imidazo-, and pyrrolopyrimidines and -pyridines. J Med Chem42:833-48 (1999) [PubMed] Article Chorvat, RJ; Bakthavatchalam, R; Beck, JP; Gilligan, PJ; Wilde, RG; Cocuzza, AJ; Hobbs, FW; Cheeseman, RS; Curry, M; Rescinito, JP; Krenitsky, P; Chidester, D; Yarem, JA; Klaczkiewicz, JD; Hodge, CN; Aldrich, PE; Wasserman, ZR; Fernandez, CH; Zaczek, R; Fitzgerald, LW; Huang, SM; Shen, HL; Wong, YN; Chien, BM; Arvanitis, A Synthesis, corticotropin-releasing factor receptor binding affinity, and pharmacokinetic properties of triazolo-, imidazo-, and pyrrolopyrimidines and -pyridines. J Med Chem42:833-48 (1999) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Corticotropin-releasing factor receptor 1 | |||
| Name: | Corticotropin-releasing factor receptor 1 | ||
| Synonyms: | CRF-R | CRF-R2 Alpha | CRF1 | CRFR | CRFR1 | CRFR1_HUMAN | CRH-R 1 | CRHR | CRHR1 | Corticotropin releasing factor receptor 1 | Corticotropin-releasing factor receptor 1 (CRF-1) | Corticotropin-releasing factor receptor 1 (CRF1) | Corticotropin-releasing hormone receptor 1 | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 50744.31 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P34998 | ||
| Residue: | 444 | ||
| Sequence: |
| ||
| BDBM50074455 | |||
| n/a | |||
| Name | BDBM50074455 | ||
| Synonyms: | Bis-(2-methoxy-ethyl)-[2,5,6-trimethyl-7-(2,4,6-trimethyl-phenyl)-7H-pyrrolo[2,3-d]pyrimidin-4-yl]-amine | CHEMBL169083 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C24H34N4O2 | ||
| Mol. Mass. | 410.5524 | ||
| SMILES | COCCN(CCOC)c1nc(C)nc2n(c(C)c(C)c12)-c1c(C)cc(C)cc1C |(6.09,-3.06,;7.42,-2.29,;8.76,-3.06,;10.1,-2.29,;11.42,-3.06,;12.75,-2.29,;12.75,-.75,;14.08,.02,;14.08,1.58,;11.42,-4.6,;10.1,-5.37,;10.1,-6.9,;8.74,-7.69,;11.42,-7.67,;12.75,-6.9,;14.22,-7.37,;15.12,-6.13,;16.66,-6.15,;14.22,-4.89,;14.68,-3.42,;12.75,-5.37,;14.85,-8.78,;16.38,-8.95,;17.29,-7.7,;17.01,-10.35,;16.1,-11.6,;16.71,-13.02,;14.57,-11.44,;13.94,-10.03,;12.4,-9.88,)| | ||
| Structure | 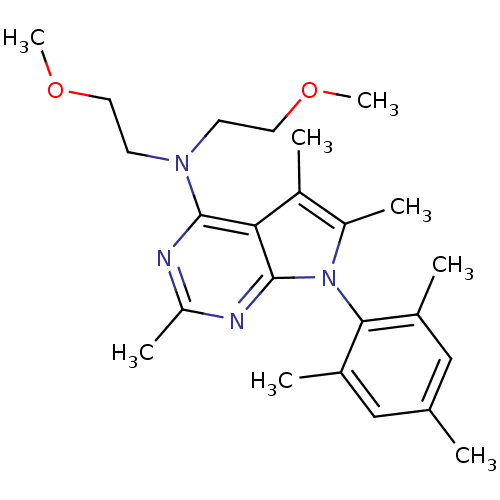
| ||