| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50075933 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_209092 |
|---|
| Ki | 5.1±n/a nM |
|---|
| Citation |  Zhang, M; Bailey, DL; Bastian, JA; Briggs, SL; Chirgadze, NY; Clawson, DK; Denney, ML; Gifford-Moore, DS; Harper, RW; Johnson, LM; Klimkowski, VJ; Kohn, TJ; Lin, HS; McCowan, JR; Richett, ME; Sall, DJ; Smith, AJ; Smith, GF; Snyder, DW; Takeuchi, K; Utterback, BG; Yan, SC Dibasic benzo[b]thiophene derivatives as a novel class of active site directed thrombin inhibitors: 2. Sidechain optimization and demonstration of in vivo efficacy. Bioorg Med Chem Lett9:775-80 (1999) [PubMed] Zhang, M; Bailey, DL; Bastian, JA; Briggs, SL; Chirgadze, NY; Clawson, DK; Denney, ML; Gifford-Moore, DS; Harper, RW; Johnson, LM; Klimkowski, VJ; Kohn, TJ; Lin, HS; McCowan, JR; Richett, ME; Sall, DJ; Smith, AJ; Smith, GF; Snyder, DW; Takeuchi, K; Utterback, BG; Yan, SC Dibasic benzo[b]thiophene derivatives as a novel class of active site directed thrombin inhibitors: 2. Sidechain optimization and demonstration of in vivo efficacy. Bioorg Med Chem Lett9:775-80 (1999) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM50075933 |
|---|
| n/a |
|---|
| Name | BDBM50075933 |
|---|
| Synonyms: | 2-[4-(2-Amino-ethyl)-phenyl]-3-(3-methoxy-4-pyrrolidin-1-ylmethyl-benzyl)-benzo[b]thiophen-6-ol | CHEMBL164246 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H32N2O2S |
|---|
| Mol. Mass. | 472.642 |
|---|
| SMILES | COc1cc(Cc2c(sc3cc(O)ccc23)-c2ccc(CCN)cc2)ccc1CN1CCCC1 |
|---|
| Structure | 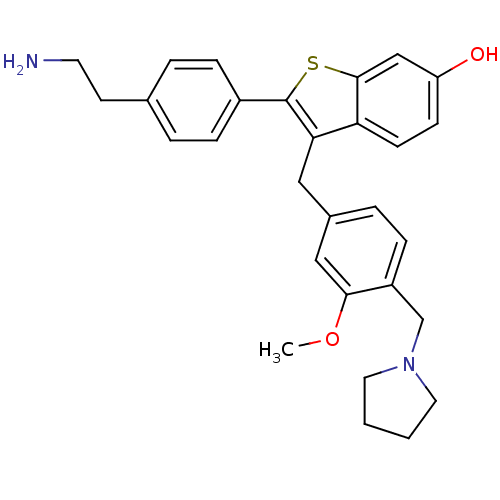
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhang, M; Bailey, DL; Bastian, JA; Briggs, SL; Chirgadze, NY; Clawson, DK; Denney, ML; Gifford-Moore, DS; Harper, RW; Johnson, LM; Klimkowski, VJ; Kohn, TJ; Lin, HS; McCowan, JR; Richett, ME; Sall, DJ; Smith, AJ; Smith, GF; Snyder, DW; Takeuchi, K; Utterback, BG; Yan, SC Dibasic benzo[b]thiophene derivatives as a novel class of active site directed thrombin inhibitors: 2. Sidechain optimization and demonstration of in vivo efficacy. Bioorg Med Chem Lett9:775-80 (1999) [PubMed]
Zhang, M; Bailey, DL; Bastian, JA; Briggs, SL; Chirgadze, NY; Clawson, DK; Denney, ML; Gifford-Moore, DS; Harper, RW; Johnson, LM; Klimkowski, VJ; Kohn, TJ; Lin, HS; McCowan, JR; Richett, ME; Sall, DJ; Smith, AJ; Smith, GF; Snyder, DW; Takeuchi, K; Utterback, BG; Yan, SC Dibasic benzo[b]thiophene derivatives as a novel class of active site directed thrombin inhibitors: 2. Sidechain optimization and demonstration of in vivo efficacy. Bioorg Med Chem Lett9:775-80 (1999) [PubMed]