| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-mannosidase |
|---|
| Ligand | BDBM50525016 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1895273 (CHEMBL4397308) |
|---|
| Ki | 500±n/a nM |
|---|
| Citation |  R�squez-Cuadro, R; Matsumoto, R; Ortega-Caballero, F; Nanba, E; Higaki, K; Garc�a Fern�ndez, JM; Ortiz Mellet, C Pharmacological Chaperones for the Treatment of ?-Mannosidosis. J Med Chem62:5832-5843 (2019) [PubMed] Article R�squez-Cuadro, R; Matsumoto, R; Ortega-Caballero, F; Nanba, E; Higaki, K; Garc�a Fern�ndez, JM; Ortiz Mellet, C Pharmacological Chaperones for the Treatment of ?-Mannosidosis. J Med Chem62:5832-5843 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-mannosidase |
|---|
| Name: | Alpha-mannosidase |
|---|
| Synonyms: | Alpha-mannosidase | Alpha-mannosidase, heavy subunit | Alpha-mannosidase, light subunit | JBM | Jbalpha-man | MANA_CANEN |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 108565.93 |
|---|
| Organism: | Canavalia ensiformis |
|---|
| Description: | ChEMBL_109557 |
|---|
| Residue: | 959 |
|---|
| Sequence: | MKYNTGAGTVPEQLNVHLVPHSHDDVGWLKTVDQYYVGSENYIQEACVENVLDSVVMSLQ
RDPNRKFVFGEMAFFHRWWLEQTPETKELKLVKAGQLEFVNGGWCMHDEATTHYIDMIDH
TTLGHRFLQEQFNKIPRAGWQIDPFGHSAVQGYLLGAELGFDSVHFARIDYQDREKRKGE
KSLEVVWRGSKTFGSSAQIFANAFPGHYGPPNGFNFEVRNNFVPLQDDPRLFDTNVEERV
QNFLDAALTQAKLTRTNHLMWTMGDDFQYQYAESWFKQMDKLLHHVNKDGRVNALYSTPS
LYTEAKNAANQTWPLKIDDYFPYADGRNAYWTGFYTSRMLSGYYLATRHSGFFAGKKSTK
YHAFDLADALGIAQHHDAVSGTAKQHTTNDYAKRLALGASKAEAVVSSSLACLTSKQSAD
QCSAPASAFSQCHLFNISYCPPTESSLPDDKSLVVVVYNPLGWSRNEIVRIPVNDANLVV
KDSSGNKLEVQYVEMDDVTANLRSFYVKYWSLFKASVPPLGWSTYFISEATGKGTRNALT
LSQKGETLNIGPGDLKMSFSSLTGQLKRMYNSKTGVDLPIQQNYLWYESSEGDFSDYQAS
GAYLFRPNGQPPPHTVSRSSVTRVTRGPLVDEVHQKFNSWISQVTRLYKDKDHAEIEFTI
GPIPTDDGVGKEVITRMTSTMATNKEFYTDSNGRDFLKRVRDYREDWPLEVTQPVAGNYY
PLNLGLYTKDEKSEFSVLVDRATGGASIKDGEVELMLHRRTLRDDGRGVGEPLDEQVCMN
KEYTCEGLTVRGNYYLSIHKPAGGSRWRRTTGQEIYSPMLLAFTQENMENWKSSHSTKAY
AMDPNYSLPPSVALITLEELDDGLVLLRLAHLYEPSEDAEYSTLTKVELKKLFATQKLEE
LREVSLSANQEKSEMKKMKWSVEGDNEQEPQAVRGGPVSNADFVVELGPMEIRTFLLQF
|
|
|
|---|
| BDBM50525016 |
|---|
| n/a |
|---|
| Name | BDBM50525016 |
|---|
| Synonyms: | CHEMBL4457217 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H29N5O5 |
|---|
| Mol. Mass. | 371.432 |
|---|
| SMILES | [H][C@]12COC(=O)N1[C@H](NCCCCCCCCCN=[N+]=[N-])[C@@H](O)[C@@H](O)[C@@H]2O |r,@:6| |
|---|
| Structure | 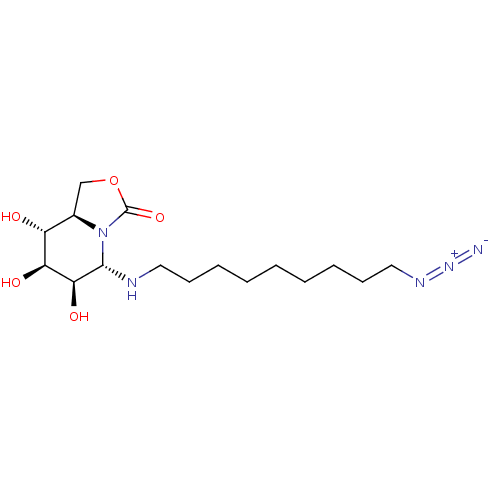
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 R�squez-Cuadro, R; Matsumoto, R; Ortega-Caballero, F; Nanba, E; Higaki, K; Garc�a Fern�ndez, JM; Ortiz Mellet, C Pharmacological Chaperones for the Treatment of ?-Mannosidosis. J Med Chem62:5832-5843 (2019) [PubMed] Article
R�squez-Cuadro, R; Matsumoto, R; Ortega-Caballero, F; Nanba, E; Higaki, K; Garc�a Fern�ndez, JM; Ortiz Mellet, C Pharmacological Chaperones for the Treatment of ?-Mannosidosis. J Med Chem62:5832-5843 (2019) [PubMed] Article