| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM12658 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_48476 (CHEMBL663038) |
|---|
| IC50 | 350±n/a nM |
|---|
| Citation |  Marlowe, CK; Sinha, U; Gunn, AC; Scarborough, RM Design, synthesis and structure-activity relationship of a series of arginine aldehyde factor Xa inhibitors. Part 1: structures based on the (D)-Arg-Gly-Arg tripeptide sequence. Bioorg Med Chem Lett10:13-6 (2000) [PubMed] Marlowe, CK; Sinha, U; Gunn, AC; Scarborough, RM Design, synthesis and structure-activity relationship of a series of arginine aldehyde factor Xa inhibitors. Part 1: structures based on the (D)-Arg-Gly-Arg tripeptide sequence. Bioorg Med Chem Lett10:13-6 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Coagulation factor X | F10 | FA10_BOVIN | Factor Xa (fXa) |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 54498.45 |
|---|
| Organism: | Bos taurus |
|---|
| Description: | ChEMBL_1364435 |
|---|
| Residue: | 492 |
|---|
| Sequence: | MAGLLHLVLLSTALGGLLRPAGSVFLPRDQAHRVLQRARRANSFLEEVKQGNLERECLEE
ACSLEEAREVFEDAEQTDEFWSKYKDGDQCEGHPCLNQGHCKDGIGDYTCTCAEGFEGKN
CEFSTREICSLDNGGCDQFCREERSEVRCSCAHGYVLGDDSKSCVSTERFPCGKFTQGRS
RRWAIHTSEDALDASELEHYDPADLSPTESSLDLLGLNRTEPSAGEDGSQVVRIVGGRDC
AEGECPWQALLVNEENEGFCGGTILNEFYVLTAAHCLHQAKRFTVRVGDRNTEQEEGNEM
AHEVEMTVKHSRFVKETYDFDIAVLRLKTPIRFRRNVAPACLPEKDWAEATLMTQKTGIV
SGFGRTHEKGRLSSTLKMLEVPYVDRSTCKLSSSFTITPNMFCAGYDTQPEDACQGDSGG
PHVTRFKDTYFVTGIVSWGEGCARKGKFGVYTKVSNFLKWIDKIMKARAGAAGSRGHSEA
PATWTVPPPLPL
|
|
|
|---|
| BDBM12658 |
|---|
| n/a |
|---|
| Name | BDBM12658 |
|---|
| Synonyms: | 4-[({[(1S)-4-carbamimidamido-1-[(4-nitrophenyl)carbamoyl]butyl]carbamoyl}methyl)carbamoyl]-4-[(2S,3S)-3-methyl-2-(phenylformamido)pentanamido]butanoic acid hydrochloride | Bz-Ile-Glu-Gly-Arg-pNA | Chromogenic Substrate S-2222 | L-Argininamide, N-benzoyl-L-isoleucyl-L-alpha-glutamylglycyl-N-(4-nitrophenyl)-, monohydrochloride | benzoyl-Ile-Glu-Gly-Arg-p-nitroanilide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H43N9O9 |
|---|
| Mol. Mass. | 697.7387 |
|---|
| SMILES | [#6]-[#6]-[#6@H](-[#6])-[#6@H](-[#7]-[#6](=O)-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-c1ccc(cc1)-[#7+](-[#8-])=O |r| |
|---|
| Structure | 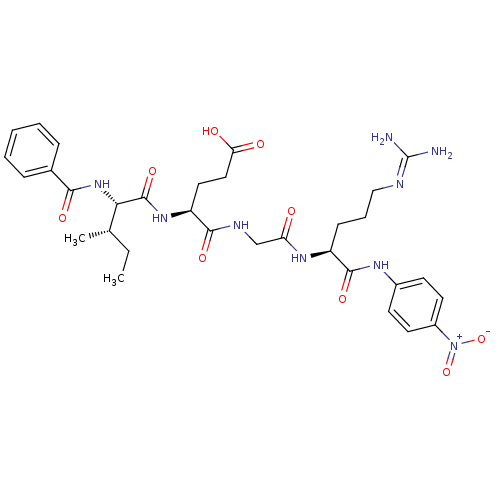
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Marlowe, CK; Sinha, U; Gunn, AC; Scarborough, RM Design, synthesis and structure-activity relationship of a series of arginine aldehyde factor Xa inhibitors. Part 1: structures based on the (D)-Arg-Gly-Arg tripeptide sequence. Bioorg Med Chem Lett10:13-6 (2000) [PubMed]
Marlowe, CK; Sinha, U; Gunn, AC; Scarborough, RM Design, synthesis and structure-activity relationship of a series of arginine aldehyde factor Xa inhibitors. Part 1: structures based on the (D)-Arg-Gly-Arg tripeptide sequence. Bioorg Med Chem Lett10:13-6 (2000) [PubMed]