| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prolyl endopeptidase |
|---|
| Ligand | BDBM50051539 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1908755 (CHEMBL4411113) |
|---|
| IC50 | 12±n/a nM |
|---|
| Citation |  Kilpel�inen, TP; Tyni, JK; Lahtela-Kakkonen, MK; Etel�inen, TS; My�h�nen, TT; Wall�n, EAA Tetrazole as a Replacement of the Electrophilic Group in Characteristic Prolyl Oligopeptidase Inhibitors. ACS Med Chem Lett10:1635-1640 (2019) [PubMed] Article Kilpel�inen, TP; Tyni, JK; Lahtela-Kakkonen, MK; Etel�inen, TS; My�h�nen, TT; Wall�n, EAA Tetrazole as a Replacement of the Electrophilic Group in Characteristic Prolyl Oligopeptidase Inhibitors. ACS Med Chem Lett10:1635-1640 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prolyl endopeptidase |
|---|
| Name: | Prolyl endopeptidase |
|---|
| Synonyms: | 3.4.21.26 | PE | PPCE_PIG | PREP | Post-proline cleaving enzyme |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 80758.04 |
|---|
| Organism: | Sus scrofa |
|---|
| Description: | n/a |
|---|
| Residue: | 710 |
|---|
| Sequence: | MLSFQYPDVYRDETAIQDYHGHKVCDPYAWLEDPDSEQTKAFVEAQNKITVPFLEQCPIR
GLYKERMTELYDYPKYSCHFKKGKRYFYFYNTGLQNQRVLYVQDSLEGEARVFLDPNILS
DDGTVALRGYAFSEDGEYFAYGLSASGSDWVTIKFMKVDGAKELPDVLERVKFSCMAWTH
DGKGMFYNAYPQQDGKSDGTETSTNLHQKLYYHVLGTDQSEDILCAEFPDEPKWMGGAEL
SDDGRYVLLSIREGCDPVNRLWYCDLQQESNGITGILKWVKLIDNFEGEYDYVTNEGTVF
TFKTNRHSPNYRLINIDFTDPEESKWKVLVPEHEKDVLEWVACVRSNFLVLCYLHDVKNT
LQLHDLATGALLKIFPLEVGSVVGYSGQKKDTEIFYQFTSFLSPGIIYHCDLTKEELEPR
VFREVTVKGIDASDYQTVQIFYPSKDGTKIPMFIVHKKGIKLDGSHPAFLYGYGGFNISI
TPNYSVSRLIFVRHMGGVLAVANIRGGGEYGETWHKGGILANKQNCFDDFQCAAEYLIKE
GYTSPKRLTINGGSNGGLLVATCANQRPDLFGCVIAQVGVMDMLKFHKYTIGHAWTTDYG
CSDSKQHFEWLIKYSPLHNVKLPEADDIQYPSMLLLTADHDDRVVPLHSLKFIATLQYIV
GRSRKQNNPLLIHVDTKAGHGAGKPTAKVIEEVSDMFAFIARCLNIDWIP
|
|
|
|---|
| BDBM50051539 |
|---|
| n/a |
|---|
| Name | BDBM50051539 |
|---|
| Synonyms: | (S)-4-phenyl-1-(2-(pyrrolidine-1-carbonyl)pyrrolidin-1-yl)butan-1-one | 4-Phenyl-1-[(S)-2-(pyrrolidine-1-carbonyl)-pyrrolidin-1-yl]-butan-1-one | 4-Phenyl-1-[2-(pyrrolidine-1-carbonyl)-pyrrolidin-1-yl]-butan-1-one | CHEMBL289651 | SUAM-1221 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H26N2O2 |
|---|
| Mol. Mass. | 314.4219 |
|---|
| SMILES | O=C(CCCc1ccccc1)N1CCC[C@H]1C(=O)N1CCCC1 |r| |
|---|
| Structure | 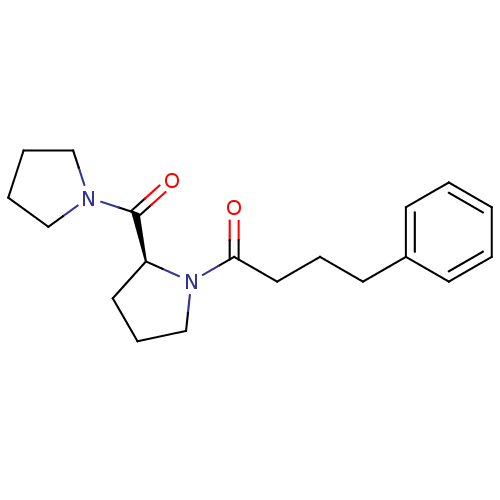
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kilpel�inen, TP; Tyni, JK; Lahtela-Kakkonen, MK; Etel�inen, TS; My�h�nen, TT; Wall�n, EAA Tetrazole as a Replacement of the Electrophilic Group in Characteristic Prolyl Oligopeptidase Inhibitors. ACS Med Chem Lett10:1635-1640 (2019) [PubMed] Article
Kilpel�inen, TP; Tyni, JK; Lahtela-Kakkonen, MK; Etel�inen, TS; My�h�nen, TT; Wall�n, EAA Tetrazole as a Replacement of the Electrophilic Group in Characteristic Prolyl Oligopeptidase Inhibitors. ACS Med Chem Lett10:1635-1640 (2019) [PubMed] Article