| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Dual specificity protein kinase CLK2 |
|---|
| Ligand | BDBM50335638 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1911749 (CHEMBL4414195) |
|---|
| IC50 | 4.0±n/a nM |
|---|
| Citation |  Bestgen, B; Kufareva, I; Seetoh, W; Abell, C; Hartmann, RW; Abagyan, R; Le Borgne, M; Filhol, O; Cochet, C; Lomberget, T; Engel, M 2-Aminothiazole Derivatives as Selective Allosteric Modulators of the Protein Kinase CK2. 2. Structure-Based Optimization and Investigation of Effects Specific to the Allosteric Mode of Action. J Med Chem62:1817-1836 (2019) [PubMed] Article Bestgen, B; Kufareva, I; Seetoh, W; Abell, C; Hartmann, RW; Abagyan, R; Le Borgne, M; Filhol, O; Cochet, C; Lomberget, T; Engel, M 2-Aminothiazole Derivatives as Selective Allosteric Modulators of the Protein Kinase CK2. 2. Structure-Based Optimization and Investigation of Effects Specific to the Allosteric Mode of Action. J Med Chem62:1817-1836 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Dual specificity protein kinase CLK2 |
|---|
| Name: | Dual specificity protein kinase CLK2 |
|---|
| Synonyms: | 2.7.12.1 | CDC-like kinase 2 | CDC-like kinase 2 (CLK2) | CDC2-like kinase 2 (CLK2) | CLK2 | CLK2_HUMAN |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 60128.79 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P49760 |
|---|
| Residue: | 499 |
|---|
| Sequence: | MPHPRRYHSSERGSRGSYREHYRSRKHKRRRSRSWSSSSDRTRRRRREDSYHVRSRSSYD
DRSSDRRVYDRRYCGSYRRNDYSRDRGDAYYDTDYRHSYEYQRENSSYRSQRSSRRKHRR
RRRRSRTFSRSSSQHSSRRAKSVEDDAEGHLIYHVGDWLQERYEIVSTLGEGTFGRVVQC
VDHRRGGARVALKIIKNVEKYKEAARLEINVLEKINEKDPDNKNLCVQMFDWFDYHGHMC
ISFELLGLSTFDFLKDNNYLPYPIHQVRHMAFQLCQAVKFLHDNKLTHTDLKPENILFVN
SDYELTYNLEKKRDERSVKSTAVRVVDFGSATFDHEHHSTIVSTRHYRAPEVILELGWSQ
PCDVWSIGCIIFEYYVGFTLFQTHDNREHLAMMERILGPIPSRMIRKTRKQKYFYRGRLD
WDENTSAGRYVRENCKPLRRYLTSEAEEHHQLFDLIESMLEYEPAKRLTLGEALQHPFFA
RLRAEPPNKLWDSSRDISR
|
|
|
|---|
| BDBM50335638 |
|---|
| n/a |
|---|
| Name | BDBM50335638 |
|---|
| Synonyms: | 5-(3-Chlorophenylamino)benzo[c][2,6]naphthyridine-8-carboxylic acid | 5-[(3-chlorophenyl)amino]benzo[c][2,6]naphthyridine-8-carboxylic acid | CHEMBL1230165 | US8168651, Compound TABLE 16.17 | US9062043, Table 13, Compound 17 | US9062043, Table 20b, Compound 12 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H12ClN3O2 |
|---|
| Mol. Mass. | 349.77 |
|---|
| SMILES | OC(=O)c1ccc2c(c1)nc(Nc1cccc(Cl)c1)c1ccncc21 |
|---|
| Structure | 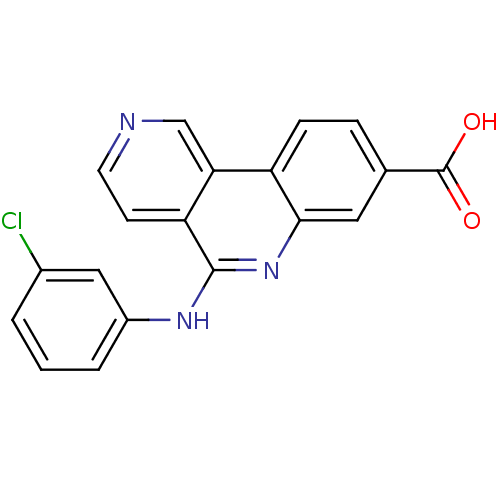
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bestgen, B; Kufareva, I; Seetoh, W; Abell, C; Hartmann, RW; Abagyan, R; Le Borgne, M; Filhol, O; Cochet, C; Lomberget, T; Engel, M 2-Aminothiazole Derivatives as Selective Allosteric Modulators of the Protein Kinase CK2. 2. Structure-Based Optimization and Investigation of Effects Specific to the Allosteric Mode of Action. J Med Chem62:1817-1836 (2019) [PubMed] Article
Bestgen, B; Kufareva, I; Seetoh, W; Abell, C; Hartmann, RW; Abagyan, R; Le Borgne, M; Filhol, O; Cochet, C; Lomberget, T; Engel, M 2-Aminothiazole Derivatives as Selective Allosteric Modulators of the Protein Kinase CK2. 2. Structure-Based Optimization and Investigation of Effects Specific to the Allosteric Mode of Action. J Med Chem62:1817-1836 (2019) [PubMed] Article