| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM50079652 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_31350 (CHEMBL641571) |
|---|
| Ki | 1300±n/a nM |
|---|
| Citation |  Harada, H; Asano, O; Hoshino, Y; Yoshikawa, S; Matsukura, M; Kabasawa, Y; Niijima, J; Kotake, Y; Watanabe, N; Kawata, T; Inoue, T; Horizoe, T; Yasuda, N; Minami, H; Nagata, K; Murakami, M; Nagaoka, J; Kobayashi, S; Tanaka, I; Abe, S 2-Alkynyl-8-aryl-9-methyladenines as novel adenosine receptor antagonists: their synthesis and structure-activity relationships toward hepatic glucose production induced via agonism of the A(2B) receptor. J Med Chem44:170-9 (2001) [PubMed] Harada, H; Asano, O; Hoshino, Y; Yoshikawa, S; Matsukura, M; Kabasawa, Y; Niijima, J; Kotake, Y; Watanabe, N; Kawata, T; Inoue, T; Horizoe, T; Yasuda, N; Minami, H; Nagata, K; Murakami, M; Nagaoka, J; Kobayashi, S; Tanaka, I; Abe, S 2-Alkynyl-8-aryl-9-methyladenines as novel adenosine receptor antagonists: their synthesis and structure-activity relationships toward hepatic glucose production induced via agonism of the A(2B) receptor. J Med Chem44:170-9 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | A2A adenosine receptor (hA2A) | AA2AR_HUMAN | ADENOSINE A2 | ADENOSINE A2a | ADORA2 | ADORA2A | Adenosine A2A receptor (A2AAR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44716.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29274 |
|---|
| Residue: | 412 |
|---|
| Sequence: | MPIMGSSVYITVELAIAVLAILGNVLVCWAVWLNSNLQNVTNYFVVSLAAADIAVGVLAI
PFAITISTGFCAACHGCLFIACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGTR
AKGIIAICWVLSFAIGLTPMLGWNNCGQPKEGKNHSQGCGEGQVACLFEDVVPMNYMVYF
NFFACVLVPLLLMLGVYLRIFLAARRQLKQMESQPLPGERARSTLQKEVHAAKSLAIIVG
LFALCWLPLHIINCFTFFCPDCSHAPLWLMYLAIVLSHTNSVVNPFIYAYRIREFRQTFR
KIIRSHVLRQQEPFKAAGTSARVLAAHGSDGEQVSLRLNGHPPGVWANGSAPHPERRPNG
YALGLVSGGSAQESQGNTGLPDVELLSHELKGVCPEPPGLDDPLAQDGAGVS
|
|
|
|---|
| BDBM50079652 |
|---|
| n/a |
|---|
| Name | BDBM50079652 |
|---|
| Synonyms: | (E)-1-[(R)-2-(2-Hydroxy-ethyl)-piperidin-1-yl]-3-(2-phenyl-pyrazolo[1,5-a]pyridin-3-yl)-propenone | (R)-1-(2-(2-hydroxyethyl)piperidin-1-yl)-3-(2-phenylpyrazolo[1,5-a]pyridin-3-yl)prop-2-en-1-one | 1-[2-(2-Hydroxy-ethyl)-piperidin-1-yl]-3-(2-phenyl-pyrazolo[1,5-a]pyridin-3-yl)-propenone | CHEMBL440115 | FK-453 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H25N3O2 |
|---|
| Mol. Mass. | 375.4635 |
|---|
| SMILES | OCC[C@H]1CCCCN1C(=O)\C=C\c1c(nn2ccccc12)-c1ccccc1 |
|---|
| Structure | 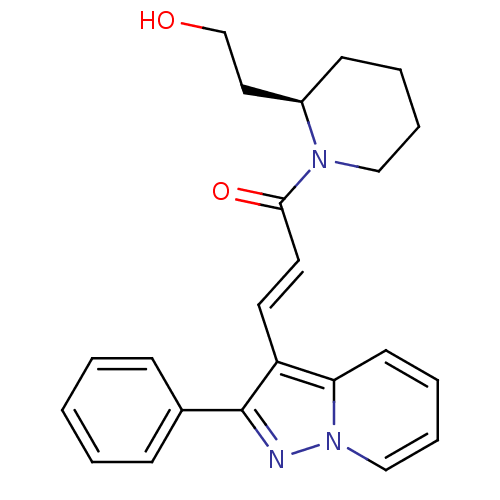
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Harada, H; Asano, O; Hoshino, Y; Yoshikawa, S; Matsukura, M; Kabasawa, Y; Niijima, J; Kotake, Y; Watanabe, N; Kawata, T; Inoue, T; Horizoe, T; Yasuda, N; Minami, H; Nagata, K; Murakami, M; Nagaoka, J; Kobayashi, S; Tanaka, I; Abe, S 2-Alkynyl-8-aryl-9-methyladenines as novel adenosine receptor antagonists: their synthesis and structure-activity relationships toward hepatic glucose production induced via agonism of the A(2B) receptor. J Med Chem44:170-9 (2001) [PubMed]
Harada, H; Asano, O; Hoshino, Y; Yoshikawa, S; Matsukura, M; Kabasawa, Y; Niijima, J; Kotake, Y; Watanabe, N; Kawata, T; Inoue, T; Horizoe, T; Yasuda, N; Minami, H; Nagata, K; Murakami, M; Nagaoka, J; Kobayashi, S; Tanaka, I; Abe, S 2-Alkynyl-8-aryl-9-methyladenines as novel adenosine receptor antagonists: their synthesis and structure-activity relationships toward hepatic glucose production induced via agonism of the A(2B) receptor. J Med Chem44:170-9 (2001) [PubMed]